Bacterial gene has key role in Legionnaires’ disease
A large-scale study of bacterial DNA samples had uncovered the significance of a gene in causing human infections.
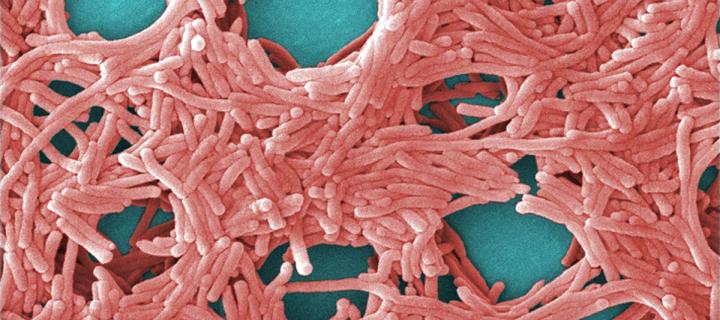
A key gene in the bacteria that cause Legionnaires’ disease helps give rise to this severe form of pneumonia, a study led by scientists from the Roslin Institute has found.
The gene enables the bacteria to resist attack from the body’s immune system in at least two ways, and helps the bug survive in the lungs during infection, researchers found.
Insights from the research could inform development of therapies for Legionnaires’ disease, which is a growing global public health problem.
Widespread study
To better understand which genes in the bacteria are important for human disease, researchers analysed and compared the genetic code of more than 900 samples of Legionella pneumophila bacteria, which causes the disease, obtained from archives of patient samples and water sources such as cooling towers.
The gene known as lag-1, which has previously been linked to infection, likely has a major role in causing disease in people, the large-scale study confirmed.
The analyses showed how different subtypes of L. pneumophila have evolved, and confirmed that some subtypes are more likely to cause severe infections.
Evading the immune system
Tests in blood plasma and serum shed light on how the lag-1 gene helps resist attack from the body’s immune system.
The gene results in a modification of the bacterial cell surface preventing recognition by molecules generated by an immune system process, known as the classic complement pathway.
Further tests involving blood immune cells known as neutrophils showed that neutrophils were prevented from working to engulf the invading cells.
Studies of mice found that infection with a lag-1-positive strain led to higher levels of bacteria in the lung compared to a strain without lag-1.
Our study pinpoints a bacterial gene with a major role in Legionnaires’ disease and highlights ways in which this impacts on the body’s immune response.
Our insights could inform the design of therapies to prevent the bacteria from resisting the response to infection, at a time when a steady global rise in infection cases is a cause for concern.
This aids our understanding of the processes that give rise to Legionnaires’ disease, and offers routes to better managing infection.
The study, published in Nature Communications, was conducted in collaboration with the Scottish Microbiology Reference Laboratory, the Charite Univeritatsmedizin Berlin, and Health Protection Scotland, and funded by the Chief Scientists Office Scotland, Wellcome, and MRC.
** The Roslin Institute receives strategic investment funding from Biotechnology and Biological Sciences Research Council and it is part of the University of Edinburgh’s Royal (Dick) School of Veterinary Studies. **
Related links
Royal (Dick) School of Veterinary Studies


