News 2018
An archive of IGMM News from 2018
Women in Cell Biology Award for Pleasantine Mill

Pleasantine Mill honoured with the 2019 Women in Cell Biology Early Career Medal: December 2018
Reverse transcriptase inhibitors in Aicardi-Goutières syndrome
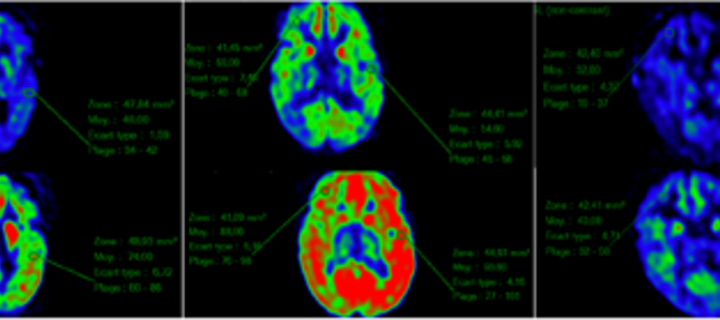
A study into the use of reverse transcriptase inhibitors for the treatment of Aicardi-Goutières syndrome. December 2018.
Our student co-organises a national conference

Laura Gomez-Cuadrado, a PhD student from the Cancer Research UK Edinburgh Centre, was a member of the organising committee of a BACR conference in London: December 2018
Generation Scotland - Leading the way in biobank data access for research

Generation Scotland is leading the way in biobank data access for research. December 2018
Mutations in a DNA methyltransferase cause microcephalic dwarfism
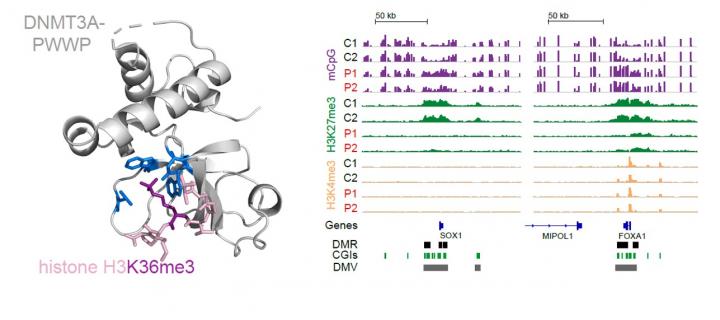
Interplay between DNA methylation and Polycomb at key developmental regulators helps determine organism size in mammals: November 2018
Donation to support bowel cancer research

Family and friends of Dennis Baron abseiled Barns Ness Lighthouse to raise money for bowel cancer research in Edinburgh: November 2018
Synthesis of palladium activated analogue of anticancer drug irinotecan
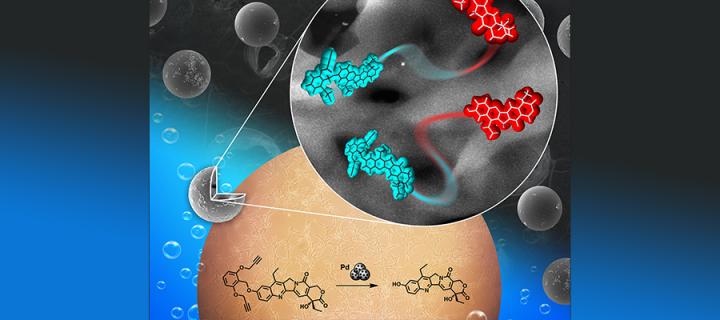
Cancer Research UK Edinburgh Centre scientists synthesise bioorthogonally activated precursor of a potent topoisomerase I inhibitor: November 2018
Success for IGMM PhD student at the Bristol Science Film Festival

Lana Woolford, a PhD student at the Cancer Research UK Edinburgh Centre, has become the Regional Winner of the Bristol Science Film Festival: November 2018
Type 1 Interferon Biomarker Study in Autoimmune Disease.

Joint funding initiative Connect Immune supports research into interferon role in autoimmunity. November 2018
Celebrating Nick Hastie’s legacy

Hastie's Career Advancement Fund will support talented early-career scientists at IGMM: November 2018
Chancellor's Award for David FitzPatrick

David FitzPatrick honoured with Chancellor's Award for Impact at Holyrood Palace
PCD Awareness Event 2018

Researchers, families and advocates discuss research, diagnostics and the patient and family perspective for primary ciliary dyskinesia (PCD): October 2018
XD challenge 2018
Undergraduate and graduate students from physics, chemistry, informatics backgrounds join IGMM scientists to tackle challenges in genetics and biomedicine: October 2018
New study assesses the effects of postmastectomy radiotherapy on quality of life in women with intermediate-risk breast cancer
Cancer Research UK Edinburgh Centre clinicians participate in clinical trial that provides important data on the consequences of postmastectomy radiotherapy in women with intermediate-risk breast cancer: October 2018
Ovarian cancer drug delays relapse

Experts from the University of Edinburgh’s Cancer Research UK Edinburgh Centre led the UK arm of an important ovarian cancer drug study: October 2018
HiP-HoP: A new inter-disciplinary approach to predict complex 3D genome folding

Gilbert and colleagues describe a new method for prediction of the 3 dimensional folding of chromatin in a Molecular Cell paper: October 2018
Epigenetic Predictors of Health and Lifestyle

Centre for Genomic and Experimental Medicine scientists develop DNA-based predictors of a range of health and lifestyle factors: October 2018
Antibiotic may selectively kill dangerous skin cancer cells
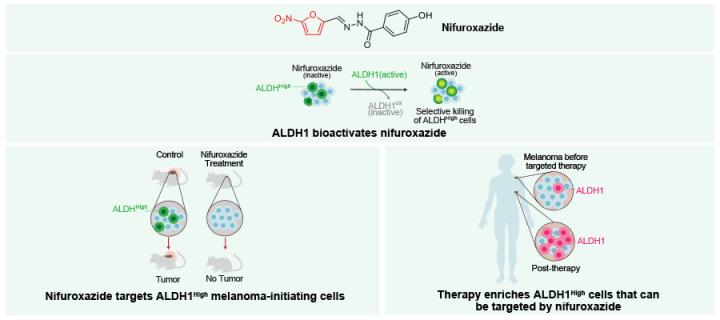
Patton and IGMM colleagues demonstrate potential use for nifuroxazide in melanoma: October 2018
HGU Hosts European Genome and Epigenome Integrity Conference

International scientific meeting of Chromatin3D training network: September 2018
£1.5m to fund Brain Tumour Centre of Excellence

Cancer Research UK Edinburgh Centre researchers have received £1.5 million for research to better understand brain tumours, one of the hardest types of cancer to treat: August 2018
Genetics Society event

Genetics Society event, in collaboration with the IGMM, at the Royal Botanic Garden Edinburgh, Edinburgh Festival Fringe, 18-26: August 2018
New Cross-Disciplinary Fellows to use big data in fight against disease

The beginning of the 21st century has seen enormous advances in science and technology that have exponentially increased our ability to gather data about the world around us, but science has yet to take full advantage of this mountain of data: August 2018
New Partnership to Develop Gene Therapy for Cystic Fibrosis

Centre for Genomic and Experimental medicine researchers are teaming up with leading figures from industry to advance development of a gene therapy for cystic fibrosis: August 2018
Edinburgh Super-resolution Imaging Consortium Summer School 2018

The fifth annual summer school of the Edinburgh Super-Resolution Imaging Consortium (ESRIC) took place at the MRC Institute of Genetics and Molecular Medicine from the 16th - 20th July. It was attended by 32 students who came from as far afield as Canada and Australia to learn from leaders in the field and access state of the art technologies: August 2018
Science Insights work experience week

It’s been another successful year for the Science Insights work experience week: August 2018
IGMM researchers in Edinburgh Festival Fringe

IGMM staff and students perform in a musical about antimicrobial resistance: August 2018
New regulatory mechanism of p53 stability revealed
![Representation of a complex between DNA and the protein p53 (By Thomas Splettstoesser [CC BY-SA 3.0]).](https://www.ed.ac.uk/sites/default/files/styles/panel_breakpoints_theme_uoe_mobile_1x/public/thumbnails/image/p53.jpg?itok=KF5O2RVy)
Cancer Research UK Edinburgh Centre scientists discover novel mechanism of regulation of p53 – an important tumour suppressor protein: July 2018
Researcher to reach new heights in charity skydive

PhD student will skydive to raise funds for ovarian cancer research: July 2018
Palladium mediated catalysis works in preclinical mouse model
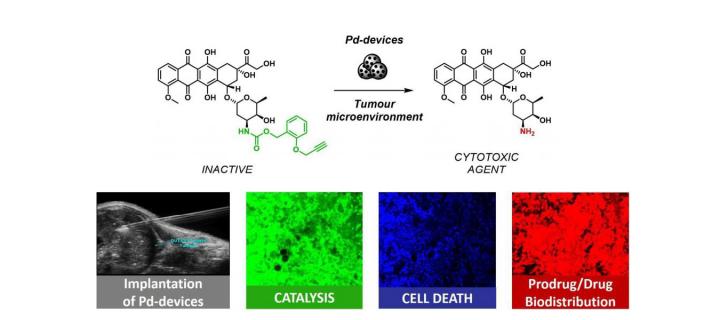
Using advanced preclinical mouse model of prostate cancer, Cancer Research UK Edinburgh Centre scientists demonstrate potential utility of palladium nanoparticles for localised drug delivery in cancer therapy: July 2018
Tours, Talks and Tea Party - people-powered genetic research

For the MRC Festival of Medical Research, the MRC Human Genetics Unit celebrated research findings that public participation in Generation Scotland has made possible: June 2018
Professor Ian Jackson, Inaugural Lecture: Marvellous melanocytes

Ian Jackson Inaugural Lecture - first at IGMM: June 2018
Cancer fighting effects of aspirin revealed in bowel tumour study

Regular use of aspirin is known to reduce a person’s risk of developing colon cancer but the drug’s tumour fighting properties have not been well understood: June 2018
Development of new inhibitors of oncogenic receptor tyrosine kinases
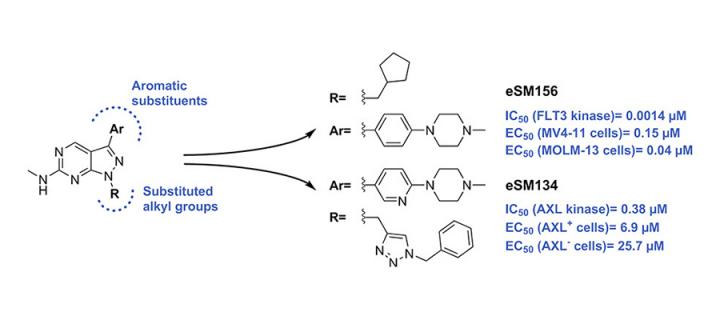
Cancer Research UK Edinburgh Centre scientists use cutting-edge approaches to develop novel inhibitors of oncogenic kinases: May 2018
Week of events promotes staff and student wellbeing

The Institute’s annual Health and Wellbeing Week took place in May, with staff and students being encouraged to take time out of their day to focus on improving their health and wellbeing: May 2018
Ground-breaking collaboration to establish how well cancer medicines really work

Dr Peter Hall from the Cancer Research UK Edinburgh Centre assumes the role of Clinical Lead on the Lothian Cancer Data Intelligence (LCDI) Collaborative Project: May 2018
£53m funding boost for MRC Human Genetics Unit

The MRC Human Genetics Unit has received a £53 million funding boost from the Medical Research Council (MRC), supporting research at the Unit for the next five years: May 2018
Bowel disease study points to new therapies for lifelong conditions

Treatments for incurable bowel conditions may be a step closer following the discovery of a key molecule associated with disease flare-ups. Researchers say the finding helps to explain the underlying cause of disorders such as Crohn’s disease and ulcerative colitis. The discovery could also lead to new tests to help doctors monitor patients’ condition and help them to tailor treatments accordingly: May 2018
Cancer Research UK Staff Meet Edinburgh Researchers

Shop workers get behind the scenes at the Cancer Research UK Edinburgh Centre: May 2018
Dr David Hunt awarded Emerging Leader Prize

Congratulations to Dr David Hunt, who is based between the MRC Institute of Genetics and Molecular Medicine and the Centre for Clinical Brain Sciences at the University of Edinburgh, for winning the Medical Research Foundation’s first Emerging Leaders Prize: April 2018
PhD students explore the role of scientists in industry

The MRC Institute of Genetics and Molecular Medicine hosted the first “Science at the Interface to Industry” 3 day workshop: April 2018
Successes at the UK Autophagy Network Meeting

Cancer Centre scientists won prizes at the 4th UK Autophagy Network Meeting: April 2018
Edinburgh scientists leading groundbreaking research to help predict bowel cancer risk

Scientists in Edinburgh are leading a groundbreaking study that could help doctors and scientists identify patients at risk of developing bowel cancer and find new treatments for the disease: April 2018
3 IGMM researchers awarded ERC Advanced Grants

Crow, Jackson and Ralston awarded grants to study interferonopathies, brain size and Paget’s disease: April 2018
Professor Charlie Gourley named NHS Hero by the Herald

Professor Charlie Gourley’s important contributions to Scotland's healthcare featured by a leading national newspaper: April 2018
Multimillion pound boost for Edinburgh scientists to tackle brain tumours

SCIENTISTS in Edinburgh are set to benefit from over two million pounds to find new ways to tackle brain tumours: March 2018
Cross-Disciplinary Fellowships (XDF) Programme

Up to 8 fellows who have acquired strong data analytical and/or computational skills from their PhD studies in physics, mathematics, computer science, engineering, or similar are to be recruited: Closing date 8 March 2018
Light Sheet Meeting at the MRC IGMM

The Advanced Imaging Resource showcased this powerful imaging technique: March 2018
Cancer patients’ pain can be eased by simple bedside chart

Patients with cancer could benefit from a new approach to manage their pain: March 2018
Dr Liz Patton - Woman of the Week in Stylist magazine

Visible Women initiative – celebrating women making a difference to society: March 2018
New directions for IGMM-industry collaboration
Bioinformatics workshop with Canon Medical Research Europe Ltd: March 2018
First Modern Apprentice at the IGMM

IGMM Design and Publication Modern Apprentice Assistant: Connor Warnock: March 2018
Kindlin-1 regulates mammary tumour metastasis

A study led by Professor Valerie Brunton from the Cancer Research UK Edinburgh Centre demonstrated that Kindlin-1 promotes pulmonary breast cancer metastasis: March 2018
Prof Chris Ponting becomes FRSE

Professor Chris Ponting has been elected as a Fellow of the Royal Society of Edinburgh: February 2018
Cancer drugs could transform the lives of children with blood vessel defects

Drugs normally used to treat cancer could reduce the disfigurements of thousands of children born with life-threatening blood vessel defects: February 2018
Killing cancer cells using palladium nanoparticles
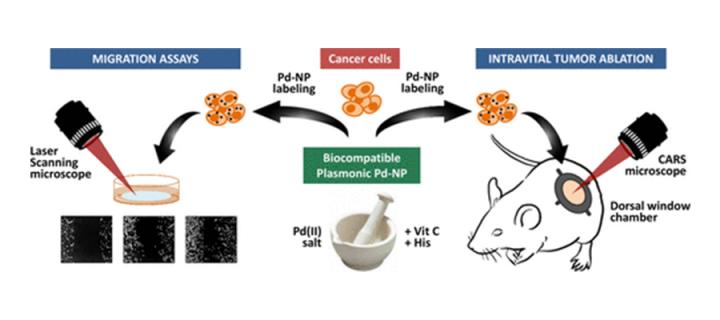
Cancer Research UK Edinburgh Centre scientists develop a scalable method for preparing palladium nanoparticles suitable for the phothothermal ablation of cancer cells: February 2018
Cancer Centre scientists link new gene to autophagy

A study led by Dr Simon Wilkinson from the CRUK Edinburgh Centre identifies cell-cycle progression gene 1 (CCPG1) as an autophagy gene: February 2018
MRC IGMM Health, Safety and Wellbeing Strategy Launched

The MRC Institute of Genetics and Molecular Medicine launched its 2018-2022 Health, Safety and Wellbeing Strategy on Monday 5 February 2018
Enhancer link to developmental disorders
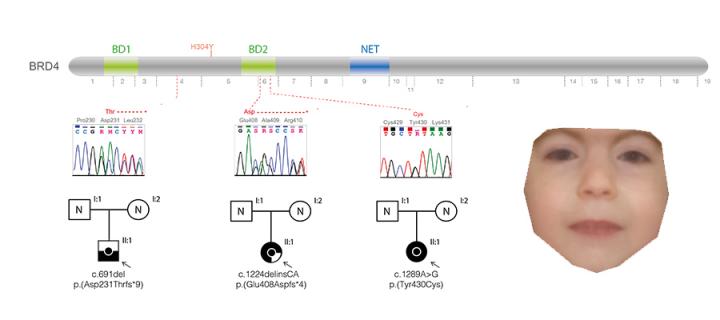
A new genetic cause of Cornelia de Lange–like syndrome improves our understanding of how our genome controls our development: January 2018
Scottish Genomes Partnership hosts Cabinet Secretary for Health visit to Edinburgh Genomics

Genome sequencing is being used to help families with an undiagnosed rare disease: January 2018
Genetic variation linked to problem-solving abilities

A DNA study of Generation Scotland participants reveals that genes are a key to problem-solving powers: January 2018
ESRIC symposium 2018

The third annual ESRIC Symposium was hosted by the IGMM: January 2018
Broughton High School Careers Fair

MRC IGMM participates annually, engaging with local pupils and parents: January 2018
Feature: Behind the scenes with Joseph Flower, our work experience videographer

Joseph has created several videos that showcase life at the IGMM. Réka Nagy writes about him in this feature: January 2018
Feature: The many faces of Mairead Bermingham

Mairead is a versatile researcher, teacher, and she often represents the IGMM at public engagement events. Réka Nagy celebrates her achievements in this feature: January 2018
Professor Margaret Frame receives OBE

New Year Honour for IGMM Director: January 2018

