Cryopreservation of chicken genetic resources
The preservation of rare chicken breeds and research flocks is important for both food security and research. Learn more about how the NARF is cryopreserving chicken genetic resources.
Cryopreserving research chicken lines at the NARF
In the next few years, the NARF intends to safeguard research lines by cryopreserving each line. Gene-edited and transgenic chicken lines that have not been used for research for extended periods, will only be maintained as cryopreserved materials, not live flocks. This reduces the number of live animals that need to be maintained annually for research purposes, addressing the principles of Replacement, Refinement and Reduction of animal use in research. Recently, lines that had not been used for many years have been cryopreserved and are now only available as cryopreserved genetic material, the Notch Reporter, Lox-STOP-lox and Talpid3.
Improvements in PGC and embryonic gonad collection and storage have enhanced the NARF’s ability to cryopreserve research chicken lines. The facility is in the process of creating a biobank to maintain the genetic resources of the unique research lines held at the NARF. A biobank allows for the storage and protection of these lines, by mitigating against loss of resources in the event of disease outbreak or genetic bottlenecks in closed populations. The development of surrogate host chickens enables lines to be brought back from cryopreserved materials if they become useful for future research projects.
Cryopreserving chicken genetic material
Poultry meat and eggs are the most widely consumed animal source of protein. Globally, there are more than 1600 chicken breeds (FAO)1 many of which have been selectively bred for traits important for food production, like increased egg laying or growth. This global diversity of chicken breeds is also a result of adaption of chickens to different environmental conditions around the world. These genetically unique chicken breeds display distinctive traits that have aided their survival, such as heat tolerance or resistance to specific diseases2.
The chicken is also an important animal model used for research, with breeding flocks maintained in research institutes. These research chicken lines are essential for research into poultry production, but also for biomedical and basic research.
Chicken breeds or research lines can be lost as a result of unpredictable events, like disease outbreaks or genetic bottlenecks in small populations. The maintenance costs of flocks that are not currently useful to farmers or researchers, can also put them at risk, leading to the loss of rare chicken breeds and research lines due to a lack of resources.
The cryopreservation of chicken breeds has been a challenge, as it has only been possible to cryopreserve male genetic material via sperm collection and storage3. Female genetic material could not be cryopreserved due to the characteristics of the chicken egg. This is limiting as only some of the genetic diversity of a chicken breed could be preserved.
Sperm collection and cryopreservation is a time-consuming process, and the viability of frozen chicken sperm can vary. To regenerate a flock from cryopreserved sperm involves inseminating females with a different genetic background, therefore necessitating multiple generations to bring back the breed.
Methods to preserve both male and female chicken genetic resources that can be used to efficiently regenerate a breed are needed.
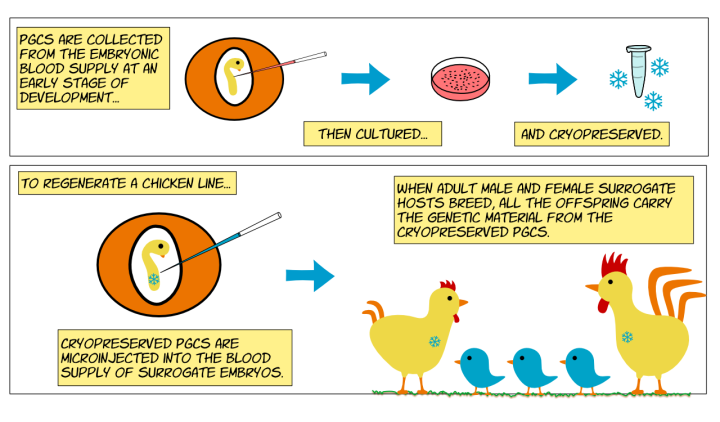
Recent progress
Key developments have facilitated our ability to cryopreserve chicken genetic resources and regenerate breeds or lines from frozen genetic material. Specifically, improvements to the collection and in vitro culture of Primordial Germ Cells (PGCs)4,5. PGCs are stem cells that give rise to sperm or eggs, and they can be collected from the blood supply of early-stage chicken embryos and grown in culture. These PGCs can be collected from both male and female early-stage embryos and cryopreserved5.
The ability to harvest and grow PGCs in culture has aided the production of novel genetically altered (GA) chicken lines. This is because PGCs can be genetically modified in culture, and then reintroduced into early-stage embryos where they migrate to the testes or eggs of the developing embryo. These methods have been used to create a transgenic surrogate host chicken line (iCaspase9) that allows for the chemical ablation of endogenous PGCs in the embryo6.
A transgenic surrogate host chicken line vastly improves our ability to regenerate chicken lines or breeds from frozen PGCs. This is because when cryopreserved PGCs are introduced into a surrogate host embryo, their endogenous PGCs can be ablated. This results in their gametes (sperm and eggs) containing only the introduced genetic material when the embryos mature into adults. Furthermore, PGCs that have been collected from both male and female embryos can be introduced back into both male and female surrogate host embryos. This reduces the time required to regenerate a line from cryopreserved materials6,7. See below a video of PGCs being introduced into the circulation of a developing embryo.
- Video: Introduction of Primordial Germ Cells into a developing chicken embryo
- Fertile chicken eggs can be windowed, allowing access to the developing chicken embryo. On Day 2 (15-16+ HH) of development, genetically altered Primordial Germ Cells (PGCs) or cryopreserved PGCs can be introduced by microinjection into the circulation of a surrogate host embryo, where there will migrate to the reproductive organs.
Ongoing research
PGC collection and propagation are both time-consuming and technically demanding, requiring specialist knowledge and equipment. To address this, alternative cryopreservation methods have been developed. Recently a study by Hu et al. (2022)8 showed that collecting embryonic gonads (testes and ovaries) from early-stage embryos can provide frozen genetic material that is as effective as frozen PGCs at regenerating a chicken line. This method also allows for the genetic material from many individuals to be collected and cryopreserved in a short time8.
Continuing research is further developing cryopreservation methods. To bring back a genetically diverse breed of chicken, it would be parsimonious to breed surrogate hosts that carry the genetic material from multiple donors. It is still unclear whether germ cell genotypes from multiple donors injected into a single surrogate host are transmitted equally to the offspring. Currently, the surrogate host line is maintained on a commercial brown layer background. Ongoing research is investigating whether the genetic background of the cryopreserved breed affects the viability of the embryos produced by surrogate hosts8,9.
As these are novel techniques, the viability of cryopreserved PGCs and embryonic gonads over time will be validated over the coming years.
Useful Links
Sterile surrogate host chicken lines; iCaspase9 and DDX4 KO
Genome engineering facilities at the NARF
Publications
- FAO - https://www.fao.org/poultry-production-products/production/poultry-species/chickens/en/
- Gheyas, A.A. et al. (2021) Integrated environmental and genomic analysis reveals the drivers of local adaptation in African indigenous chickens. Molecular Biology and Evolution, 38(10), 4268–4285.
- Thélie, A. et al. (2019). Chicken semen cryopreservation and use for the restoration of rare genetic resources. Poultry Science, 98(1), 447-455.
- Van De Lavoir, M. C. et al. (2006) Germline transmission of genetically modified primordial germ cells. Nature, 441, 766–769.
- Whyte, J. et al. (2015) FGF, insulin, and SMAD signaling cooperate for avian primordial germ cell self-renewal. Stem Cell Reports, 5, 1171–1182.
- Ballantyne, M. et al. (2021) Direct allele introgression into pure chicken breeds using Sire Dam Surrogate (SDS) mating. Nat. Commun. 12, 659.
- Ballantyne, M. et al. (2021) Avian primordial germ cells are bipotent for male or female gametogenesis. Frontiers in Cell and Developmental Biology, 9: 726827.
- Hu, T. et al. (2022) A low-tech, cost-effective and efficient method for safeguarding genetic diversity by direct cryopreservation of poultry embryonic reproductive cells. Elife, 11: e74036.
- NC3Rs - Cryo-storing research lines of chicken to eliminate breeding and save genetic resources.


