Stem cells FAQs
Some frequently asked questions on the topic of stem cells.
For lots more information and resources about stem cells and regenerative medicine, including information about using stem cells in the clinic and clinical trials of stem cell therapies, visit the EuroStemCell website.
What is a stem cell?
A stem cell is a type of cell which is able to do two things. Firstly, they can divide to produce a more specialised cell which is needed in the body, such as a muscle cell or a red blood cell. This process of becoming more specialised is called differentiation. Stem cells can also self-renew, which means that they can replace themselves so that your supply of stem cells doesn’t run out.
This EuroStemCell video gives you a short introduction to the world of stem cells and stem cell research.
Why do we need stem cells?
Our bodies are made up of trillions of cells. Most of these cells can live for anything from a few days to a few years before they die and need to be replaced. Stem cells provide new specialised cells to replace dying ones.
Are there different kinds of stem cells?
There are many different kinds of stem cells but some of the most important ones are:
Tissue stem cells – These cells are found in your body and provide new cells to your organs. They can usually only make a limited number of different cell types which are specific to the organ or tissue that they are found in. For example, intestinal stem cells can produce the different types of cell found in the intestine, but not other cells in other organs like nerve cells. These cells are also known as adult stem cells, but are also found in children and foetuses.
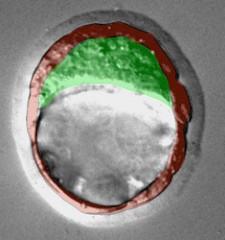
Embryonic stem cells – Embryonic stem cells are considered to be more powerful than adult stem cells as they can make any type of cell found in the body. Embryos which are a few days old, known as blastocysts, are used to make ES cells in the lab. Blastocysts have two main parts; the trophectoderm, shown in red in this image, which surrounds the blastocyst and goes on to form the placenta, and the inner cell mass, shown in green, which is found inside the blastocyst. The cells in the inner cell mass are the ones to develop into the actual organism which will eventually be born.
Embryonic stem cells are produced when the inner cell mass is removed from a blastocyst and grown in the lab. The cells can be grown indefinitely and used to make all kinds of different cells for both scientific research and, potentially, to replace dying cells in patients with diseases like Parkinson’s. However, there are ethical issues surrounding the use of embryonic stem cells as their production results in the destruction of an embryo.
Induced pluripotent stem cells – Induced pluripotent stem cells, or iPS cells, are another type of stem cell that can be made in the lab from normal adult cells, such as skin cells. Adding four additional factors into the cells allows them to go back to a similar state as embryonic stem cells. In this state they can also produce any cell type found in the body, like embryonic stem cells.
The technology for making iPS cells was developed by Professor Shinya Yamanaka’s research group at the University of Kyoto and was announced to the scientific community in 2006. It has revolutionised the fields of stem cell research and regenerative medicine. Professor Yamanaka subsequently shared the 2012 Nobel Prize for Physiology or Medicine for his work on iPS cells.
This video will tell you more about iPS cells and Professor Yamanaka’s work.
What are the advantages of iPS cells?
iPS cells could offer significant advantages over embryonic stem cells. It is now possible to make stem cells from patients with severe degenerative diseases. iPS cells from patients can then be differentiated into specialised cells, for example, iPS cells from a patient with Alzheimer’s disease could be turned into nerve cells. This can allow scientists to learn more about the disease by studying the patient’s cells in the lab.
Healthy cells can also be taken from donors or even from the patient themselves and used to produce ‘replacement’ cells to be transplanted back into the patient. This drastically increases the supply of cells and tissues available for transplant into patients. It also means that cells which are a close genetic match to the patient can be selected. Transplanting genetically matched cells into a patient reduces the severity of transplant rejection by the patient’s immune system.
iPS cells are also seen as a more ethically acceptable type of stem cell than embryonic stem cells, as no embryos are destroyed in the production of iPS cells. However, there are still ethical issues surrounding the production and use of iPS cells in the clinic.
What does pluripotent mean?
The ‘potency’ of stem cells is a measure of how many different cell types they can produce. Embryonic stem cells and iPS cells are pluripotent, as they can form all of the cell types needed to make an organism, but they cannot form cells outside of the embryo (for example. cells in the placenta). Tissue stem cells can only make a small range of cell types, limited to the organ that they are found in. This makes them multipotent.
How could stem cells help patients?
Many patients around the world have diseases where cells in their bodies don’t function properly (for example, insulin-producing cells in diabetes patients) or start to die without being replaced (such as in Alzheimer’s disease and Parkinson’s disease). Stem cells could be used to replace these defective or dying cells, restoring normal function to the organ and improving the patient’s quality of life.
The most immediate use of iPS cells which could help patients is by using iPS cells in drug discovery and testing. iPS cell technology means that iPS cells with different genetic profiles can be easily turned into different types of specialised cells or tissues and used to discover new therapeutic drugs or to check for harmful side-effects of drug candidates.
For information about the use of stem cells to treat specific diseases, such as heart disease and spinal cord injuries, visit the EuroStemCell website.
Related links
Read more about the ethical discussion surrounding iPS cells [EuroStemCell]


