The role of staphylococcal superantigen SElW
New article characterises the distribution and elusive function of the SElW superantigen in Staphylococcus aureus.
Many years of hard work have now come to fruition with our recent mBIO publication “Population Analysis of Staphylococcus aureus Reveals a Cryptic, Highly Prevalent Superantigen SElW That Contributes to the Pathogenesis of Bacteremia”. This work, led by LBEP alumni Manouk Vrieling and Stephen Tuffs, was achieved in collaboration with Dr Tim Connelly's group at the Roslin Institute and research groups from Mississippi State university and the University of Western Ontario.
Read the article abstract here:
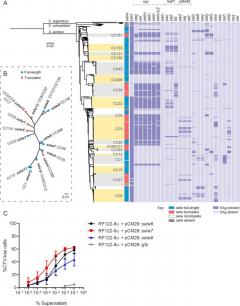
"Staphylococcal superantigens (SAgs) are a family of secreted toxins that stimulate T cell activation and are associated with an array of diseases in humans and livestock. Most SAgs produced by Staphylococcus aureus are encoded by mobile genetic elements, such as pathogenicity islands, bacteriophages, and plasmids, in a strain-dependent manner. Here, we carried out a population genomic analysis of >800 staphylococcal isolates representing the breadth of S. aureus diversity to investigate the distribution of all 26 identified SAg genes. Up to 14 SAg genes were identified per isolate with the most common gene selw (encoding a putative SAg, SElW) identified in 97% of isolates. Most isolates (62.5%) have a full-length open reading frame of selw with an alternative TTG start codon that may have precluded functional characterization of SElW to date. Here, we demonstrate that S. aureus uses the TTG start codon to translate a potent SAg SElW that induces Vβ-specific T cell proliferation, a defining feature of classical SAgs. SElW is the only SAg predicted to be expressed by isolates of the CC398 lineage, an important human and livestock epidemic clone. Deletion of selw in a representative CC398 clinical isolate, S. aureus NM001, resulted in complete loss of T cell mitogenicity in vitro, and in vivo expression of SElW by S. aureus increased the bacterial load in the liver during bloodstream infection of SAg-sensitive HLA-DR4 transgenic mice. Overall, we report the characterization of a novel, highly prevalent, and potent SAg that contributes to the pathogenesis of S. aureus infection."
For more information, you can find the paper in this link: https://mbio.asm.org/content/11/5/e02082-20.full
Full Citation:
Manouk Vrieling, Stephen W. Tuffs, Gonzalo Yebra, Marleen Y. van Smoorenburg, Joana Alves, Amy C. Pickering, Joo Youn Park, Nogi Park, David E. Heinrichs, Lindert Benedictus, Timothy Connelley, Keun Seok Seo, John K. McCormick, J. Ross Fitzgerald. mBio Oct 2020, 11 (5) e02082-20; DOI: 10.1128/mBio.02082-20

