Bacterial host-specialized fibrinogen-binding
New article characterizes the high strength binding of a cell-wall associated protein of Staphylococcus pseudintermedius to canine fibrinogen
We have just published a new article entitled “Host-specialized fibrinogen-binding by a bacterial protein promotes biofilm formation and innate immune evasion” in PLOS Pathogens. This work was led by Amy Pickering in collaboration with a number of research groups from Trinity College Dublin, Université Catholique de Louvain in Belgium, East Carolina University, and Texas A&M Health Science Center in the US.
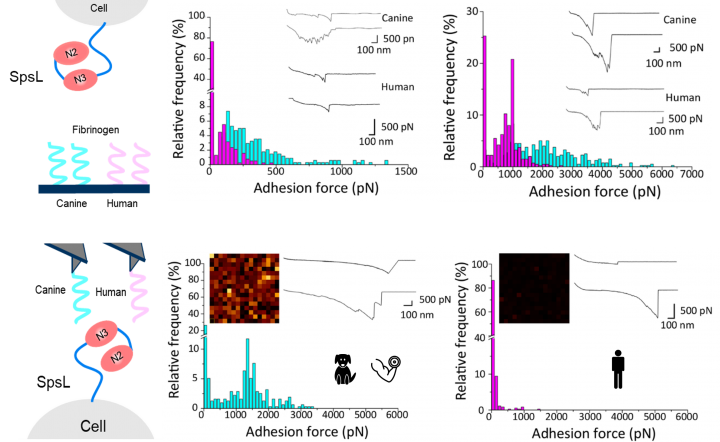
Many bacterial pathogens are specialized for a single host-species and rarely cause infections of other hosts. Our understanding of the bacterial factors underpinning host-specificity are limited. Here we demonstrate that a canine host-restricted bacterial pathogen, Staphylococcus pseudintermedius, produces a surface protein (SpsL) that has the ability to preferentially bind to canine fibrinogen with high strength. This host-specific interaction has evolved via binding to a tandem repeat region of the fibrinogen alpha-chain which is divergent among mammalian species. Importantly, we found that the strong binding interaction with canine fibrinogen promotes bacterial aggregation and biofilm formation as well as inhibiting neutrophil phagocytosis. Our findings reveal the host-adaptive evolution of a key bacterium-host interaction that promotes evasion of the host immune response.
For more information, you can find the paper in this link: https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1007816
Full Citation:
Pickering AC, Vitry P, Prystopiuk V, Garcia B, Höök M, Schoenebeck J, Geoghegan JA, Dufrȇne Y, Fitzgerald JR. (2019) Host-specialized fibrinogen-binding by a bacterial surface protein promotes biofilm formation and innate immune evasion. PLoS Pathog 15(6): e1007816. https://doi.org/10.1371/journal.ppat.1007816

