Genetic variation and RNA structure regulate microRNA biogenesis
Control of miR30c-1 levels is affected by genetic changes found in breast and gastric cancer patients: May 2017
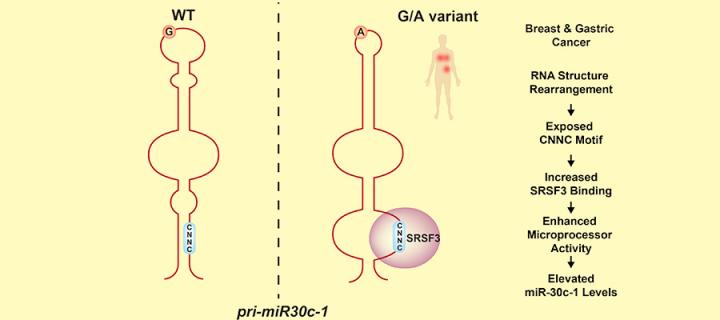
RNAs are important molecules in the cell which contain, and regulate, genetic information. MicroRNAs (miRNAs) are short non-coding RNAs that negatively regulate the expression of a large fraction of messenger RNAs (mRNAs) affecting a multitude of cellular and developmental pathways. Dysregulated miRNA expression can result in grossly aberrant gene expression leading to human disease; highlighting the need for miRNA levels to be tightly controlled. Initially harboured within long primary RNA transcripts (termed the primary miRNA), miRNAs require successive nuclear and cytoplasmic processing steps to release their mature forms. The cellular machinery that performs the nuclear and cytoplasmic processing steps are the Microprocessor complex and Dicer, respectively.
In this paper, Fernandez, Cordiner and colleagues from the MRC Human Genetics Unit at the University of Edinburgh uncovered the mechanisms by which a rare genetic variation in the terminal loop of pri-miR-30c-1 (G27 to A) found in breast and gastric cancer patients leads to increased levels of the corresponding mature miRNA. They showed that this genetic variant induces an RNA secondary structure rearrangement that facilitates binding of a trans-acting factor (SRSF3), which leads to increased processing of this primary miRNA in the nucleus, resulting in enhanced levels of the mature miRNA (see Figure). These data highlights that primary sequence determinants and RNA structure are key regulators in miRNA biogenesis. This could be of importance in human populations, whereby previously overlooked changes in the non-coding portion of the genome can influence miRNA biogenesis and result in a concurrent effect on global gene expression.
Fernandez,N.±, Cordiner,R.A.±, Young,R.S., Hug,N., Macias,S. and Cáceres,J.F. (2017) Genetic variation and RNA structure regulate microRNA biogenesis. Nat. Commun., 8, 15114 | doi: 10.1038/ncomms15114
±Joint First authors
Caceres Lab research: http://www.ed.ac.uk/mrc-human-genetics-unit/research/caceres-group


