KDM3A is a master regulator of cilia stability
Yeyati, Mill and colleagues show a critical role for the histone demethylase KDM3A in cilia stability: March 2017
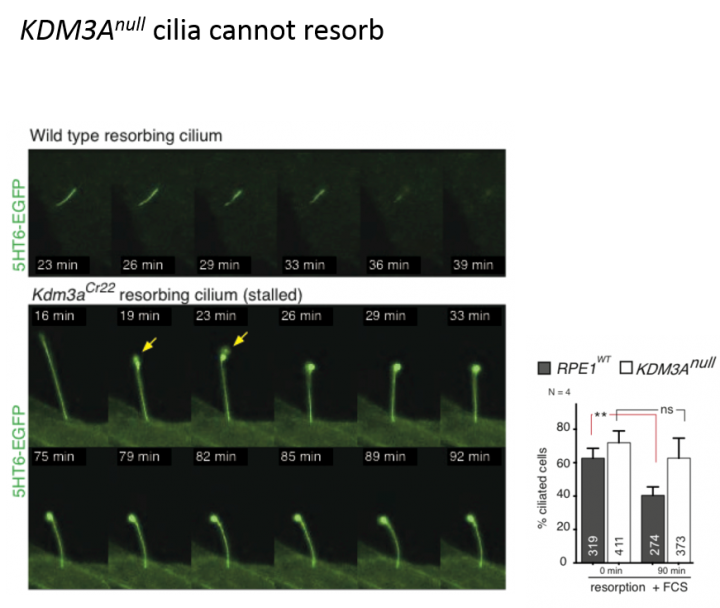
Cilia are small, microtubule-based structures found on the surface of almost every mammalian cell. They act as important cellular antennae that sense and trigger cellular responses to signals in the extracellular environment. Cells can change their behaviour in response to environmental signals by changing gene expression, organization of their cytoskeleton and cilial length. How these complex processes are tightly integrated was unclear. Patricia Yeyati, Pleasantine Mill, and other colleagues at the HGU have uncovered that the histone demethylase KDM3A, previously known to control gene expression in the nucleus, plays a key master regulatory role in merging all these inputs. Interestingly, mice with loss of function mutations in KDM3A are obese, predisposed to diabetes and males can be infertile, which are also observed in mouse models with dysfunctional cilia. In a paper published this week, they show KDM3A acts as a key linchpin in the coordination of these complex cellular processes to ensure a coherent cellular response. KDM3A modulates actin dynamics transcriptionally, as well as by directly binding actin. Cells lacking KDM3A show restricted intraflagellar transport as cilia are formed, leading to unstable cilia and accumulation of intraflagellar transport proteins.
Reference: J Cell Biol. 2017 Feb 28. pii: jcb.201607032. doi: 10.1083/jcb.201607032. [Epub ahead of print]
http://jcb.rupress.org/content/early/2017/02/27/jcb.201607032.long


