Adhesion protein that regulates gene transcription via direct signalling across the nuclear envelope
Edinburgh-led study identifies a novel function for the cell adhesion protein Mena at the nuclear membrane, where it regulates actin-nuclear lamina associations, nuclear architecture, chromatin repositioning and gene expression: March 2023
Mechanotransduction is a term that describes the ability of cells to actively sense, integrate, and convert mechanical stimuli into biochemical signals that result in intracellular changes. Defects in mechanotransduction have been implicated in the development of a wide array of diseases, ranging from muscular dystrophies and cardiomyopathies to cancer progression and metastasis.
Cell adhesion complexes – protein complexes that form with adhesion receptors in the cell membrane and mediate interactions between neighbouring cells or with molecules in the surrounding extracellular environment – and the cytoskeleton, a structure that helps cells maintain their shape and internal organisation, have well established functions as sensors of mechanical forces and key mediators of mechanotransduction.
For quite a while, researchers have known that mechanical forces are important for regulation of multiple cellular processes, including genome organisation and gene expression in the cell nucleus, but the mechanisms involved and the links between cell adhesion complexes, cytoskeletal proteins and nuclear functions remain poorly understood. A recent study led by Edinburgh Cancer Researchers at the Institute of Genetics and Cancer provided important new insights into this.
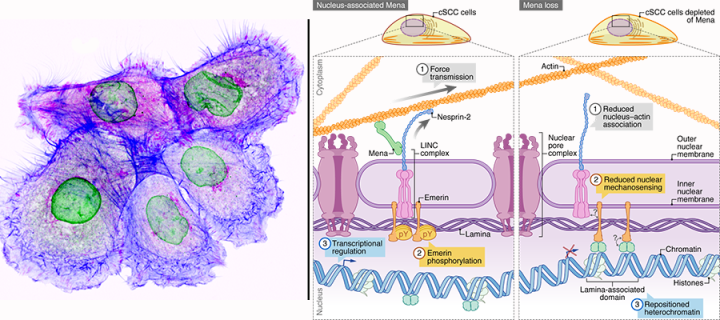
Scientists used patient-derived cutaneous squamous cell carcinoma cells (squamous cell carcinoma is a second most common form of skin cancer) to analyse components of the so-called “integrin adhesome” – a set of molecules that are associated with the structure and signalling activity of cell adhesions mediated by adhesion receptors known as integrins. They employed cutting-edge technologies, including quantitative proteomics, high- and super-resolution cell imaging and DNA sequencing, to show that a protein called Mena, which is usually found at sites of cell adhesion near the plasma membrane, has a role in transmitting force from the cytoskeleton into the nucleus of cells. They discovered that Mena interacts with the nuclear envelope protein nesprin-2 and that it regulates the morphology of the nucleus, the organisation of chromatin and gene expression. The study provides important new insights to our understanding of mechanotransduction in cancer.
The work, which represents a great team effort across multiple research labs, was driven by Dr Frederic Li Mow Chee and supervised by Prof Margaret Frame and Dr Adam Byron. It has been published by the journal Nature Communications in the article titled “Mena regulates nesprin-2 to control actin–nuclear lamina associations, trans-nuclear membrane signalling and gene expression”. The work was supported by funding from Cancer Research UK, the European Research Council, the Medical Research Council, the European Union and the Wellcome Trust.
This work establishes a connection between the adhesome component Mena and the LINC complex, through which Mena regulates actin-nuclear lamina interactions, nuclear architecture and chromatin organisation at the nuclear periphery, fine-tuning gene expression. We found that Mena regulated the expression of genes involved in cell migration and the immune response, which are very important biological processes in cancer progression.
Related Links
Article in Nature Communications: https://www.nature.com/articles/s41467-023-37021-x
Prof Margaret Frame group website: https://www.ed.ac.uk/cancer-centre/research/frame-group
Dr Adam Byron profile: https://www.ed.ac.uk/profile/adambyron
Information about skin cancer: https://www.cancerresearchuk.org/about-cancer/skin-cancer
Related Stories
- Characterisation of a nucleo-adhesome
- Loss of Integrin-Linked Kinase sensitizes breast cancer to SRC inhibitors
- Focal Adhesion Kinase controls transcription via chromatin accessibility
- Focal Adhesion Kinase activation on lipid membranes


