Drivers of endocrine therapy resistance in estrogen receptor positive breast cancer
Edinburgh researchers used a unique sample set to provide new insights into endocrine therapy resistance in estrogen receptor positive breast cancer: August 2022
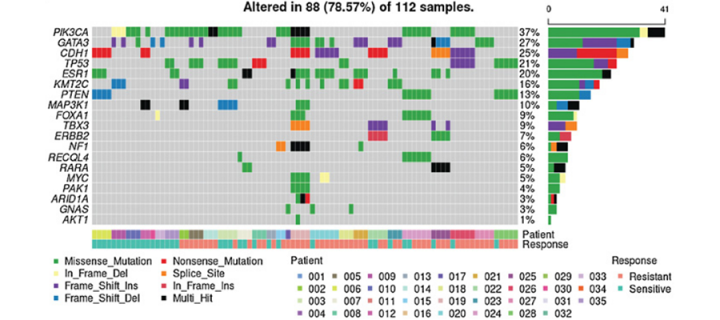
Breast cancer remains one of the leading causes of mortality in women with over 11,000 breast cancer deaths every year in the UK alone. More than 75% of breast cancers test positive for estrogen receptors (proteins that bind to estrogens), and are known as estrogen receptor positive (ER+).
Estrogens are a group of hormones that play an important role in the normal sexual and reproductive development in women. Cancer cells that are estrogen receptor positive often need estrogens to grow. Consequently, agents that reduce levels of circulating estrogens (e.g. letrozole, anastrozole, exemestane) or target the estrogene receptors (e.g. tamoxifen or fulvestrant) are commonly used in clinical practice. Over the years they have contributed hugely to improving patients outcomes. Unfortunately, not all ER+ patients respond well to these therapeutics due to a variety of reasons including intrinsic resistance resulting from natural molecular heterogeneity and cellular phenotypes that develop and evolve during treatment. Endocrine therapy resistance (ETR), sometimes also described as hormone therapy resistance, remains the biggest barrier to prolonged survival and cure for patients with ER+ breast cancer. Mechanisms of ETR are subject of intense investigations driven by hope that their understanding might result in better treatment strategies in the future.
To this end our investigators, in collaboration with researchers from the University of North Carolina in Chapel Hill, performed a detailed analysis (at DNA and RNA level) of multiple tumor samples derived from patients whose cancers were responsive to endocrine therapy, had intrinsic resistance, or developed acquired resistance. Importantly, the samples were collected from patients before treatment and at different time points during the treatment, including biological replicate samples from the same resistant tissue to assess tumor spatial heterogeneity. This approach provided valuable insights into changes happening in individual tumors over time and data on intratumor heterogeneity. The study, titled “Integrated DNA and RNA Sequencing Reveals Drivers of Endocrine Resistance in Estrogen Receptor-Positive Breast Cancer”, revealed that the mechanisms underlying ETR are multiple and characterized by diverse changes in both genetic (DNA) and transcriptomic (RNA) profiles. It indicates that individualized approach utilizing genomic and genetic biomarkers and drugs tailored to each patient might be required to overcome ETR.
The work, supervised by Dr Arran Turnbul and Professor Michael Dixon, was published recently in the journal Clinical Cancer Research. It was supported by funding from Breast Cancer Now, Lyda Henderson Fund, Chewning Family, Breast Cancer Research Foundation and Cancer Research UK.
We are deeply grateful to the patients who allowed us to sample and use their tumor samples for this study. Our work confirmed some known mechanisms of endocrine therapy resistance and identified some novel mechanisms. Importantly it has also highlighted the heterogeneity that underlies breast tumor biology.
Related Links
Article in Clinical Cancer Research: https://aacrjournals.org/clincancerres/article/doi/10.1158/1078-0432.CCR-21-3189/
Dr Arran Turnbull Group website: https://www.ed.ac.uk/cancer-centre/research/turnbull-group
Professor Michael Dixon Group website: https://www.ed.ac.uk/cancer-centre/research/dixon-group
Information about breast cancer: https://www.cancerresearchuk.org/about-cancer/breast-cancer
Related Stories
- Multi-omic machine learning predictor of breast cancer therapy response
- Loss of Integrin-Linked Kinase sensitizes breast cancer to SRC inhibitors
- Guidelines for treatment of breast cancer patients with delays in surgery due to COVID-19
- Professor David Cameron appointed BIG Chair
- In HER2 positive early breast cancer 6 months treatment with Herceptin is as good as 12 months for preventing cancer return
- HER2 drives an increased hypoxic response in breast cancer
- Distinguishing acquired resistance from dormant tumours in neoadjuvant treatment of breast cancer
- Best poster prize at the Edinburgh Breast Cancer Special Symposium

