Kidney cancer drug accepted for use within NHS Scotland and across the UK
Results of an international study with its UK component led from Edinburgh has led to approval of the first ever treatment reducing relapse after kidney cancer surgery: October 2022
On Monday the 10th of October, the Scottish Medicines Consortium (SMC; the national source of advice on the clinical and cost-effectiveness of all new medicines for NHS Scotland) advised on a new indication for the use of pembrolizumab (brand name Keytruda) within NHS Scotland. The drug has been accepted for use within NHS Scotland as a monotherapy for the adjuvant treatment of adults with renal cell carcinoma (RCC) at increased risk of recurrence following nephrectomy (surgery to remove a kidney or part of a kidney), or following nephrectomy and resection of metastatic lesions. The advice applies in the context of an approved NHS Scotland Patient Access Scheme. This SMC advice accompanies similar advice from the National Institute for Health and Care Excellence (NICE) in England and Wales (final appraisal document and draft guidance released 16th of September) and, together, will result in UK-wide NHS access to this important new treatment.
In Scotland, 937 patients were diagnosed with RCC in 2017, and the incidence of the disease has continually increased in recent decades. RCC is generally resistant to chemotherapy and radiation therapy and surgical resection is the only curative therapy. Unfortunately, about one-third of patients present with metastatic disease and have a 5-year survival rate of less than 15%. Approximately 25% of patients with localised RCC who undergo a nephrectomy have disease recurrence at distant sites, but this can rise to over 50% for those whose original RCC was larger or involved lymph nodes or blood vessels, and the greatest risk of recurrence is within 3-5 years after surgery. Before the approval of pembrolizumab, no further treatment (adjuvant treatment) was available to prevent or reduce the risk of relapse of the cancer following nephrectomy.
Following surgery, patients diagnosed with locally advanced kidney cancer live with the uncertainty and concern of possible recurrence or metastatic spread of cancer. This leads to stress and anxiety, often affecting the wellbeing of the patients and those close to them, and having detrimental effect on family life.
Pembrolizumab represents a therapeutic advancement as it is the first medicine to have shown meaningful benefit in reducing recurrence of kidney cancer following surgery in those who have intermediate or high risk of disease recurrence. Patients groups associated with charities Action Kidney Cancer and Kidney Cancer Scotland indicated that:
The availability of an adjuvant treatment reduces the stress and anxiety for patients and their families and carers caused by potential recurrence of the disease following surgery. This improves their wellbeing enabling them to continue to contribute socially and economically to society. Patients will typically be travelling some distance to a regional cancer centre for the pembrolizumab infusions. However most patients feel much better able to cope with life knowing that they are taking a treatment to prevent the cancer from recurring.
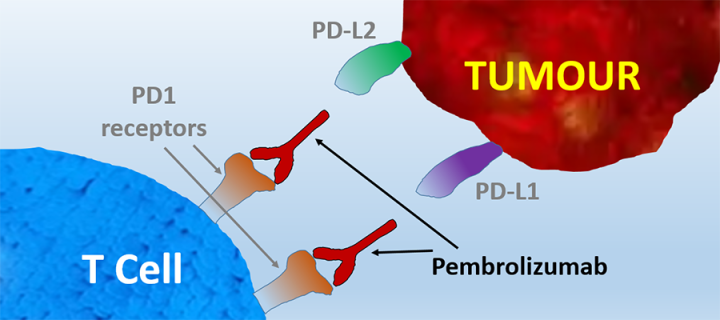
Pembrolizumab is a humanized antibody which targets the programmed cell death protein 1 (PD-1) receptor of T lymphocytes (T cells, a type of white blood cells constituting an essential part of the immune system). When functioning properly, T cells are activated and can attack tumour cells but some tumours can evade the immune system through the PD-1 pathway. The PD‑L1 and PD‑L2 ligand proteins on tumour cells can bind with PD-1 receptors on T cells to inactivate the T cells. Pembrolizumab binds to the PD-1 receptor and blocks its interaction with PD‑L1 and PD‑L2, which helps restore the immune response.
Approval of pembrolizumab was possible because of positive results from the KEYNOTE-564 study, a randomised, double-blind, phase III clinical trial that enrolled 994 patients at 213 study locations across 21 countries. In the United Kingdom, it included hospitals in Edinburgh, Glasgow, London, Manchester, Middlesbrough and Stoke-On-Trent. Some of the key results from the study were published by the New England Journal of Medicine in an article titled “Adjuvant Pembrolizumab after Nephrectomy in Renal-Cell Carcinoma” and, more recently, by Lancet Oncology in an article titled “Pembrolizumab versus placebo as post-nephrectomy adjuvant therapy for clear cell renal cell carcinoma (KEYNOTE-564): 30-month follow-up analysis of a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial”. One of our investigators, Dr Stefan Symeonides, served as the UK Chief Investigator of the KEYNOYTE-564 trial and was an author on both publications. Dr Symeonides is a Medical Oncologist at the Edinburgh Cancer Centre and a Senior Clinical Lecturer at the University of Edinburgh Institute of Genetics and Cancer. He is also Co-lead of the Edinburgh Experimental Cancer Medicine Centre responsible for early phase clinical trials of promising new cancer therapeutics.
The KEYNOYTE-564 trial has shown that pembrolizumab reduces the risk of kidney cancer coming back by more than a third, with signs that this is leading to our patients living longer and disease free. Many UK patients kindly participated in the trial and it is fantastic news that this treatment is becoming available within the UK, as the first ever treatment for reducing relapse after kidney cancer surgery
It is important to remember that pembrolizumab is not a “wonder drug”. It is not always effective and research continues to explore which people might benefit most from treatment. In addition, the treatment can cause undesired adverse effects like fatigue, severe itching, hypothyroidism or others, including the risk of severe or persistent immune side-effects. Nonetheless, it represents a significant breakthrough in treatment of kidney cancer as confirmed by UK approvals from SMC in Scotland and NICE in England and Wales, as well as its incorporation in treatment guidelines from the European Society for Medical Oncology (ESMO). Approval of pembrolizumab is a welcome news to many RCC patients, who otherwise had no treatment options and often felt abandoned by the healthcare system.
Related Links
Advice from the Scottish Medicine Consortium regarding the use of pembrolizumab (Keytruda) after nephrectomy in Renal-Cell Carcinoma:
https://www.scottishmedicines.org.uk/medicines-advice/pembrolizumab-keytruda-rcc-full-smc2479/
October 2022 decisions news release from the Scottish Medicine Consortium:
https://www.scottishmedicines.org.uk/about-us/latest-updates/october-2022-decisions-news-release/
“Pembrolizumab versus placebo as post-nephrectomy adjuvant therapy for clear cell renal cell carcinoma (KEYNOTE-564): 30-month follow-up analysis of a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial”, article in the Lancet Oncology: https://www.thelancet.com/journals/lanonc/article/PIIS1470-2045(22)00487-9/fulltext
“Adjuvant Pembrolizumab after Nephrectomy in Renal-Cell Carcinoma”, article in the New England Journal of Medicine: https://www.nejm.org/doi/full/10.1056/NEJMoa2106391
Information about KEYNOTE-564 clinical trial: https://clinicaltrials.gov/ct2/show/NCT03142334
Dr Stefan Symeonides research profile: https://www.ed.ac.uk/cancer-centre/research/symeonides-group
Information about kidney cancer: https://www.cancerresearchuk.org/about-cancer/kidney-cancer
Pembrolizumab for adjuvant treatment of renal cell carcinoma [ID3810] – NICE Guidance: https://www.nice.org.uk/guidance/indevelopment/gid-ta10693
Related Stories
Clinical trial identifies effective adjuvant therapy for patients with kidney cancer
AI - empowered personalized cancer medicine
Therapy trial offers breast cancer patients hope
Optimizing treatment for the older and frailer cancer patients
Opportunities for stratification in endometrioid ovarian carcinoma

