Licensing of a SRC/YES1 inhibitor with novel mode of action
Nuvectis Pharma, a biopharmaceutical company focused on the development of innovative precision medicines, has licensed exclusive worldwide rights to develop and commercialize a novel SRC/YES1 inhibitor discovered at the CRUK Edinburgh Centre: September 2021
SRC family kinases (SFKs), including SRC, YES1 and some other family members, are enzymes capable of phosphorylating proteins on tyrosine residues. They are ubiquitously expressed in various cell types and can also act as “scaffolds” facilitating formation of multiprotein complexes essential for various aspects of cell physiology. Structure and regulation of SFKs are relatively complex and the kinases have been implicated in multiple processes such as cell proliferation, differentiation, apoptosis, migration, and metabolism.
Abnormalities in SFKs expression level and signalling have been associated with diverse human diseases, from osteoporosis and cardiovascular diseases to chronic kidney diseases and cancer. In fact, SRC is the first oncogene ever reported and has been proved a key regulator of cancer development and progression. SRC is aberrantly activated in many cancer types, including solid tumours such as breast, colon, prostate, pancreatic and ovarian cancers, while remaining predominantly inactive in non-cancerous cells. SRC overactivation is generally associated with late-stage cancers, metastatic potential and resistance to therapies and poor clinical prognosis. In addition, amplification of the YES1 gene has also been reported in various solid tumours, including lung, head and neck, esophageal and sarcoma, and has also been described as a mechanism of acquired resistance to treatment with EGFR inhibitors that develops in a substantial percentage of patients with EGFR-mutated lung adenocarcinomas.
Although several SFKs inhibitors have been described and some of them entered clinical trials, so far the clinical utility of molecular therapeutics targeting SFKs in cancer is rather disappointing. A reason for this might be that most currently available SRC inhibitors are only able to inhibit tyrosine kinase activity of SFKs, without preventing their “scaffolding” functions. Many SFKs inhibitors also potently suppress activity of other cellular kinases, like for example members of the ABL family kinases, potentially introducing additional complications with respect to safety and utility.
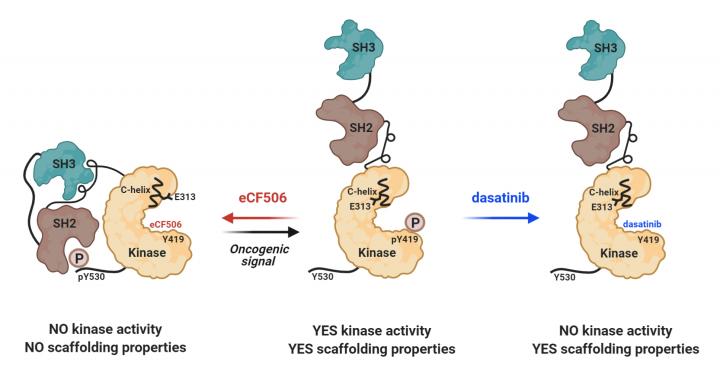
A few years ago Edinburgh researchers, led by Professors Asier Unciti-Broceta and Neil Carragher, developed a highly potent and orally bioavailable inhibitor of SRC and YES1 (called eCF506) which displays high selectivity towards some SFKs without inhibiting many other kinases present in the cells. Recently, in collaboration with colleagues from the Structural and Chemical Biology, Biological Research Center (CIB-CSIC) in Spain, they were able to show that this inhibitor locks SRC in its native inactive conformation, thereby inhibiting both enzymatic and scaffolding functions that prevent phosphorylation and complex formation with its partner Focal Adhesion Kinase (FAK). The study, titled “A novel mode of inhibiting SRC improves drug efficacy and tolerability” and published in the journal “Cancer Research”, provides evidence for unprecedented mechanism of action of eCF506. Treatment with eCF506 resulted in increased antitumor efficacy and tolerability in syngeneic murine cancer models, demonstrating significant therapeutic advantages over existing SRC/ABL inhibitors.
Following this remarkable discovery the University of Edinburgh and US biopharmaceutical company Nuvectis Pharma, Inc., have signed a licensing agreement to turn a research breakthrough into new medicines. Nuvectis Pharma has licensed exclusive worldwide rights to develop and commercialise treatments based on eCF506. The compound, which has been designated NXP900, is expected to commence IND-enabling studies (studies to secure approval to conduct the first-in-human clinical trials) in fourth quarter of 2021.
Every researcher working in this field hopes their discoveries can reach patients and save lives, and this agreement with Nuvectis promises just that, using a new way to attack cancer that has long evaded science. This is the first description of a drug made by taking a different approach of combining advanced cell-based screening with innovative medicinal chemistry to select compounds with exciting biological activities. The discovery is testament to the power of innovation in academic cancer drug discovery and especially the new approaches that we have pioneered in the Institute over many years, without spending the very large sums of money usually poured into conventional drug discovery pipelines to get new potential anti-cancer drugs to this stage. Congratulations to Asier and Neil, the Nuvectis team and Edinburgh Innovations on reaching this agreement.
We believe that NXP900’s selectivity and demonstrated potency in several animal models of cancers potentially provide a broad range of development options, both as single agent and in combination with approved drugs. We are excited to be working with Nuvectis to rapidly move NXP900 into the IND-enabling studies with the hope of commencing clinical trials for NXP900 shortly thereafter.
We are delighted to have partnered NXP900 with Nuvectis upon completion of the discovery program at the Institute of Genetics and Cancer. We are confident that Nuvectis’ skills and expertise in drug development bode well for the successful and rapid development of NXP900 and ultimately hope it becomes a much-needed new option that benefits patients with SRC/YES1 driven cancers.
We are thrilled to partner with the Institute of Genetics and Cancer at the University of Edinburgh to advance the development of NXP900, which has demonstrated outstanding preclinical activity to date. We also thank Edinburgh Innovations, the commercialization service of the university, for facilitating this licensing. We believe that with our two pipeline products, we are now one step closer to creating a diversified precision-medicine oncology-focused company with the goal of helping more people in their fight against hard-to-treat cancers.
Related Links
Article in Cancer Research: https://cancerres.aacrjournals.org/content/early/2021/08/20/0008-5472.CAN-21-0613.long
Nuvectis Pharma In-Licenses Exclusive Worldwide Rights to a Novel SRC/YES1 Inhibitor (NXP900) from the University of Edinburgh, Scotland – announcement on Yahoo! Finance: https://finance.yahoo.com/news/nuvectis-pharma-licenses-exclusive-worldwide-120000302.html
Information about the agreement on the Scottish Business News website: https://scottishbusinessnews.net/us-drug-firm-licenses-breakthrough-edinburgh-cancer-discovery/
Nuvectis Pharma, Inc. website: https://nuvectis.com/
Prof Prof. Asier Unciti-Broceta Group website: https://www.ed.ac.uk/cancer-centre/research/unciti-broceta-group
Prof Neil Carragher Group website: https://www.ed.ac.uk/cancer-centre/research/carragher-group
Edinburgh Cancer Discovery Unit site: https://www.ed.ac.uk/cancer-centre/impact-and-innovation/translational-science/edinburgh-cancer-discovery-unit-ecdu
Related Stories
- ECRC scientists develop new Src inhibitor with unique properties.
- Edinburgh Cancer Discovery Unit featured in “SLAS Discovery”.
- Synergistic anticancer inhibitor combination discovered by a novel phenotypic screen.
- Development of new inhibitors of oncogenic receptor tyrosine kinases.
- Synthesis of palladium activated analogue of anticancer drug irinotecan.
- Palladium mediated catalysis works in preclinical mouse model.
- Killing cancer cells using palladium nanoparticles.
- Gold-Triggered Uncaging Chemistry in Living Systems Opens New Frontiers for Cancer Therapy.

