Wilms’ Tumour Suppressor (WT1) - not just a transcription factor
Recent research within the MRC Human Genetics Unit (HGU) of the IGMM, has finally found a combination of roles of Wilms’ Tumour 1 (WT1) that together demonstrate its crucial importance in disease development: April 2017
The Wilms’ Tumour is the most common type of kidney cancer diagnosed in children at 3-4 years and is very rare in adults. The Wilms’ Tumour suppressor gene1 (WT1) was first cloned in 1990 as a Wilms’ Tumour suppressor, as inactivation of WT1 causes the Wilm's Tumour. Wt1 mutations may also lead to life-threatening kidney glomerulosclerosis, gonadal dysgenesis and in rare cases congenital diaphragmatic hernia (CDH) and heart disease.
WT1 is also a transcription factor, controlling the rate of transcription of genetic information from DNA to messenger RNA and therefore helping to regulate the expression of genes. It is essential for the development and homeostasis of multiple tissues, including the kidney, gonads, coronary vasculature and visceral adipose tissue.
Tumour suppression genes, such as WT1, are very important in understanding the onset and progression of cancer and in identifying potential marker genes and specific targets for the prevention and personalised treatments of cancer. WT1 has a prognostic role in a variety of types of cancer and is a promising target for immunotherapy based on its unique features.
Recent research within the MRC Human Genetics Unit (HGU) of the IGMM, has finally found a combination of roles of Wilms’ Tumour 1 (WT1) that together demonstrate its crucial importance in disease development.
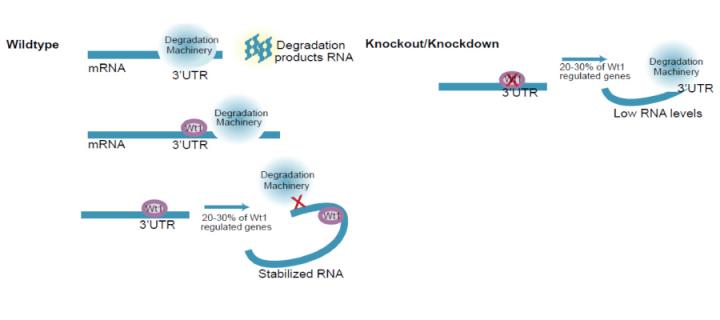
A substantial body of evidence supports post-transcriptional roles for WT1 and this research generated an unbiased Global RNA Interactome Map for endogenous WT1. This revealed preferential binding of WT1 to the three prime untranslated region (3'-UTR) of target genes and intramolecular RNA interactions in a subset of developmental regulators. As the majority of the RNA targets that depend on WT1 for their expression are not transcription targets, this research proposes that WT1 influences key developmental processes, in part through regulating mRNA turnover.
It has been widely assumed that WT1 exerts its key functions by binding to DNA and regulating transcription. However, evidence, particularly from the Hastie Lab, over the past two decades has suggested additional roles at the post-transcriptional level, from splicing through to translation by binding RNA. In spite of all this circumstantial evidence, conclusive studies have not been forthcoming, as no physiological WT1-RNA binding targets have been identified.
This study has now identified RNA targets for endogenous WT1 in both kidney and differentiated embryonic stem cells. The most striking finding is that WT1 is associated principally with intramolecular RNA interactions in the 3’ untranslated region (UTR) of the mRNA. As hypothesized, this research has shown that WT1 regulates the stability of its target RNAs through these 3’ UTR interactions.
This study not only demonstrates that WT1 is a multi-functional protein but also highlights the key role of post-transcriptional processes in development, homeostasis and disease. This is just the tip of the iceberg, as it is proposed that WT1 will be shown to regulate further steps in the cascade of gene expression. This research has also provided an important insight, providing a model that highlights the need to consider the wider context and explore multiple regulatory mechanisms to understand the role of disease causing mutations in genes.
Genes Dev. 2017 Feb 15;31(4):347-352. doi: 10.1101/gad.291500.116.

