News 2019
An archive of IGMM News from 2019
Genetic clues to retinal detachment
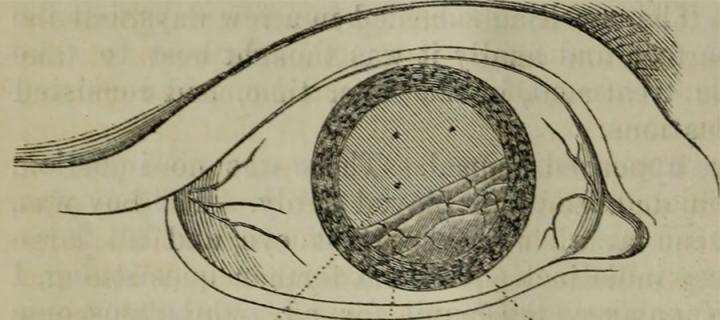
A large study has identified 6 regions of the human genome influencing risk of sight-threatening condition: December 2019
Open approach to animal research earns accolade

The University’s work on communicating how animals are used in research has won a national award: December 2019
Rarity is Common: The Genetics of Shetland

Whole genome sequencing reveals high number of ultra-rare genetic variants, particularly in gene coding and regulatory regions: December 2019
Cabinet Secretary Jeane Freeman MSP visits the MRC Human Genetics Unit

The Medical Research Council (MRC) Human Genetics Unit at The University of Edinburgh welcomed Jeane Freeman MSP, Cabinet Secretary for Health and Sport for the Scottish Government: 7 November 2019
Professor David Cameron appointed BIG Chair

Professor David Cameron becomes new Chair of the Breast International Group (BIG against breast cancer), the largest global not-for-profit network for academic breast cancer research groups: November 2019
DNA: Disease, Nature, Ancestry ─ Three billion letters of you

Professor Chris Ponting delivered a fascinating and engaging inaugural lecture at IGMM on 30 September: November 2019
Human Gut Cell Atlas – Normal Intestine and Crohn’s Disease
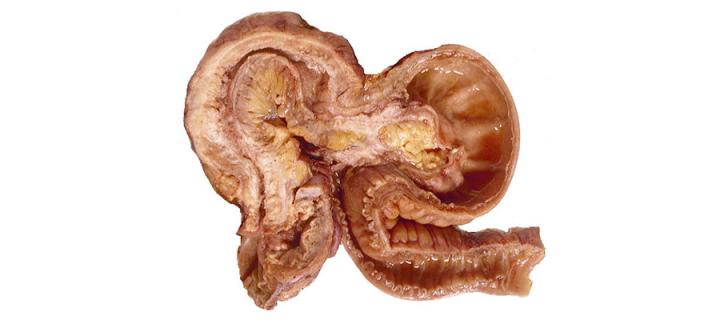
Edinburgh researchers receive support from the Helmsley Charitable Trust to provide new insights into Crohn’s disease: November 2019
House of Lords Science and Technology Committee’s Ageing Inquiry

Dr Riccardo Marioni gave evidence to a House of Lords Inquiry into Ageing. Nov 2019
XDF Programme Fellow published papers in physics and biomedicine journals

One of our XDF Programme Fellows, Dr Ava Khamseh, contributed to exciting research publications in “Physical Review B” and “Cancer Research”: November 2019
Diabetes Scotland supporters visit research labs

Professor Helen Colhoun welcomed a group of passionate fundraisers to find out more about Diabetes UK funded research at the IGMM: October 2019
IGMM welcomes the public inside for Doors Open Day

The MRC Institute of Genetics and Molecular Medicine opened its doors to the public for its fourth Doors Open Day last month, hosting almost 200 visitors: October 2019
A new role for autophagy in EGFR recycling and signalling

Cancer Research UK Edinburgh Centre scientists demonstrated that autophagy regulates recycling and signalling of Epidermal Growth Factor Receptor: October 2019
Novel cell states in melanoma progression and residual disease

Using zebrafish models to visualise cells at the site of melanoma residual disease with the potential to drive melanoma recurrence: October 2019
DNA findings suggest improved way to predict gout
Genetic data from half a million individuals informs on gout prediction and biological underpinning of high urate levels: October 2019
Drug trial offers hope of improved ovarian cancer treatments
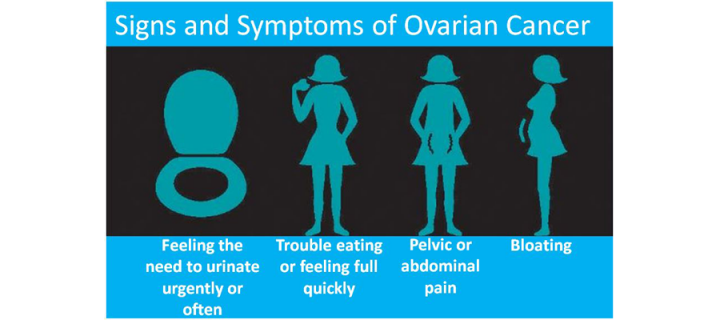
Pivotal ovarian cancer clinical trial with key contributions from Cancer Research UK Edinburgh Centre clinician: September 2019
Probing TADs in development
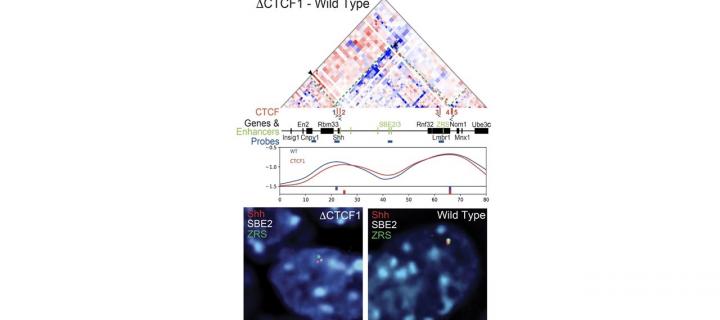
Alteration of TAD boundaries does not prevent Shh gene function or impair development: September 2019
Physicists and biologists work together to shed light on genome reorganisation in senescence
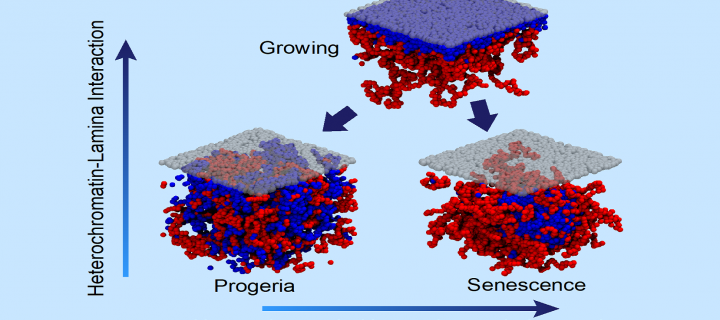
Scientists from the School of Physics and Astronomy and the MRC Human Genetics Unit at the University of Edinburgh work together to uncover mechanisms governing genome reorganisation in senescence: September 2019
Charlie Lees awarded UKRI Future Leaders Fellowship

Dr Charlie Lees has been awarded a highly prestigious URKI-funded Future Leaders Fellowship, joining the Centre for Genomic and Experimental Medicine at the University of Edinburgh in November 2019
Mathematicians and Cosmologist join Cross-Disciplinary Fellowships (XDF) Programme

IGMM welcomes newest Cross-Disciplinary Fellows: Vanessa Smer Barreto, Lyndsay Kerr and Eric Latorre Crespo: September 2019
Precious metal flecks could be catalyst for better cancer therapies

Tiny extracts of a precious metal used widely in industry could play a vital role in new cancer therapies: September 2019
Scotland’s genetic landscape echoes Dark Age populations
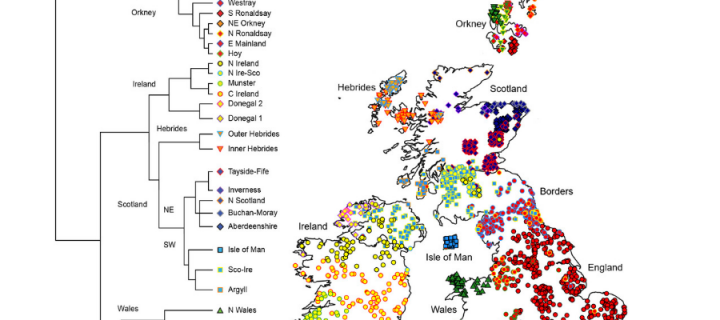
The DNA of Scottish people still contains signs of the country’s ancient kingdoms, with many apparently living in the same areas as their ancestors did more than a millennium ago, a study shows: September 2019
Activation at a distance: new evidence on enhancer-gene communication
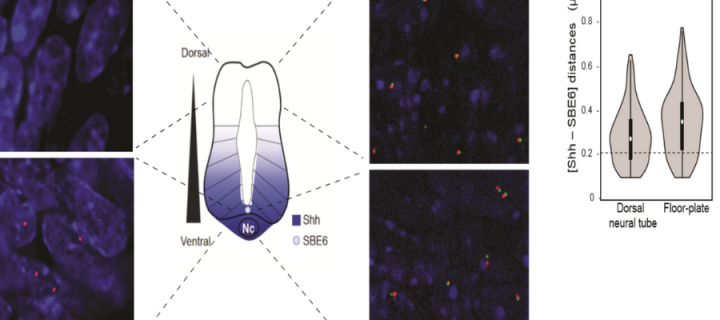
Recent findings from scientists at the MRC Human Genetics Unit have shed new light on how enhancers and genes communicate at great distance along the chromosome: September 2019
First successful research grant application with an XDF Programme Fellow as the Lead Applicant
Our XDF Programme Fellow, Dr Mattia Marenda, awarded SULSA Technology Seed Funding: September 2019
Our Academic Consultant Oncologist among the finalists of the Scottish Women's Awards 2019!

Dr Olga Oikonomidou from the Cancer Research UK Edinburgh Centre has been named for the second time as one of the finalists of the Scottish Women’s Awards: August 2019
Lothian Birth Cohort study suggests ageing process accelerated by DNA changes

MRC Human Genetics Unit and Centre for Genomic and Experimental Medicine study with University of Glasgow links somatic mutations to a general measure of biological ageing: August 2019
Edinburgh Super-resolution Imaging Consortium Summer School 2019

The sixth annual summer school of the Edinburgh Super-resolution Imaging Consortium (ESRIC) took place at the MRC Institute of Genetics and Molecular Medicine, University of Edinburgh and Heriot-Watt University 15 - 19 July 2019.
Genetic and Epigenetic Biomarkers of Protein Level in Neurological Disorders

Centre for Genomic and Experimental Medicine scientists identify genetic and epigenetic biomarkers associated with neurological disorders. July 2019
Inaugural Hastie Career Advancement Awards given to IGMM postdocs

Carmen Amador and Magda Maslon, postdoctoral researchers at the MRC Human Genetics Unit, have received inaugural awards from the Hastie Career Advancement Fund.
Cancer study to ease plight of people at advanced stages of illness
Patients with advanced cancer are to test a therapy aimed at reducing their symptoms and improving quality of life: July 2019
Genetics Garden wins Silver Medal at the RHS Chelsea Flower Show

Professor Wendy Bickmore and fellow members of the Genetics Society have proudly brought home a prestigious award by taking genetics to the general public at this year’s RHS Chelsea Flower Show.
Shining a Light on Genetic Disorders

On 18 June the public joined researchers, clinicians and patients at the MRC IGMM for ‘Shining a Light on Genetic Disorders’, part of this year’s MRC Festival of Medical Research: June 2019
Science Insights – New Insights MRC Festival of Medical Research

Over the last five years, Public Engagement staff from across the College of Medicine and Veterinary Medicine have coordinated Science Insights, a week-long summer work experience programme for S5 pupils: June 2019
£12 Million fund for scientists to perform vital brain cancer research
Scientists are set to receive over £12 million from Cancer Research UK and The Brain Tumour Charity to find new ways to treat brain tumours: June 2019
Chemists and Biologists Meet for Bioorthogonal & Bioresponsive 2019

Almost a hundred researchers from nine different countries visited IGMM to attend the Bioorthogonal & Bioresponsive 2019 symposium, 6-7 June.
Cycle Friendly Employer Award Achievement

The Institute of Genetics and Molecular Medicine (IGMM) has been awarded Cycle Friendly Employer status by Cycling Scotland.
A link between innate immune sensing and cellular senescence
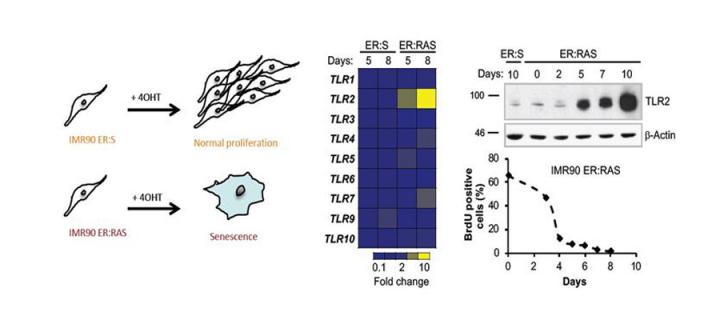
Cancer Research UK Edinburgh Centre scientists discover a cell intrinsic tumour suppressor role of the innate immune sensor Toll-like receptor 2: June 2018
Edinburgh Super-Resolution Imaging Consortium Symposium 2019

The Edinburgh Super-Resolution Imaging Consortium (ESRIC) – a partnership between University of Edinburgh and Heriott Watt University – hosted the 2019 ESRIC Symposium at IGMM on 28 May 2019.
DNA tests for patients move closer with genome analysis advance

Diseases caused by genetic changes could be detected more readily thanks to an advance in DNA analysis software.
ediRNA Meeting 2019

Bringing together RNA researchers in Edinburgh: May 2019
Reverse Transcriptase Inhibitors for Interferonopathies
A recent award of a £1.7 million MRC Experimental Medicine Challenge grant to Professor Yanick Crow will assess a novel drug treatment approach for Aicardi-Goutières syndrome. May 2019
Senescence Symposium 2019

Biomedical scientists gather in Edinburgh for Senescence Symposium: May 2019
BODYSCAPE - Living with disease through art and science

A patient engagement Sci-Art event: May 2019
Fine tuning energy production
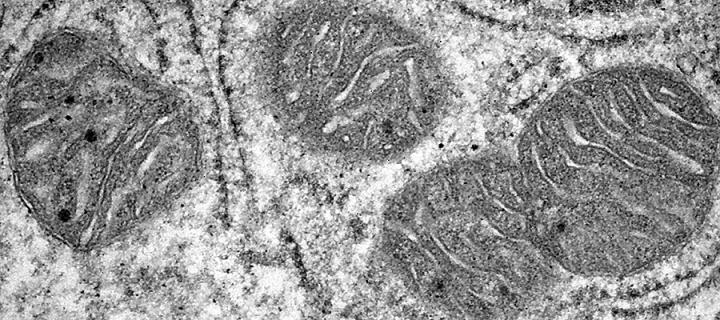
Cerox1 long non-coding RNA regulates mitochondrial activity
From Green Bench to Lab Bench

Carol Monaghan MP took a walk in the shoes of an Edinburgh health data scientist as part of the Royal Society's pairing scheme for scientists and MPs. April 2019
Breast cancer study to focus on tumour DNA in blood

Research into an advanced form of breast cancer is set to aid understanding of cancer biology. The project is focused on secondary breast cancer – an incurable form of the condition that occurs when the cancer spreads to other parts of the body: April 2019
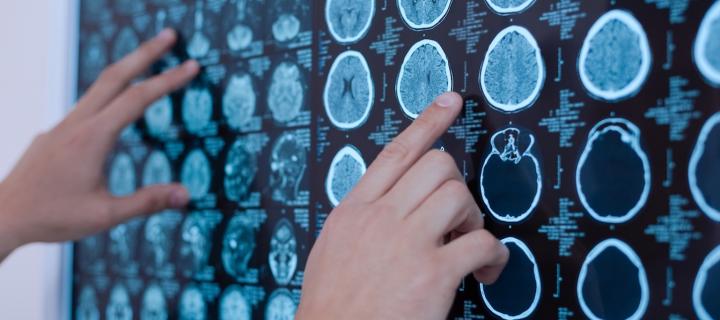
DNA modifications associated with poor lung function and COPD
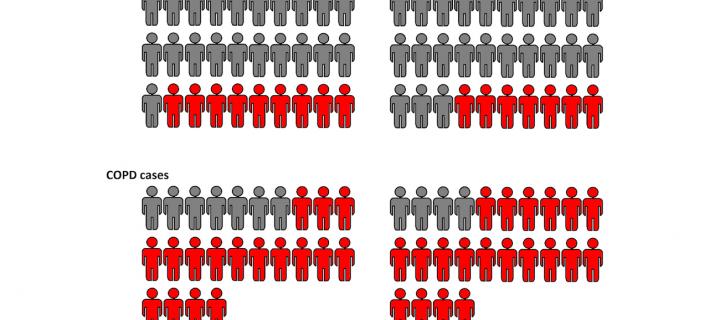
DNA is modified by the environment and genetics to affect the risk of chronic obstructive pulmonary disease. April 2019
Important new insights into the function of a key autophagy protein
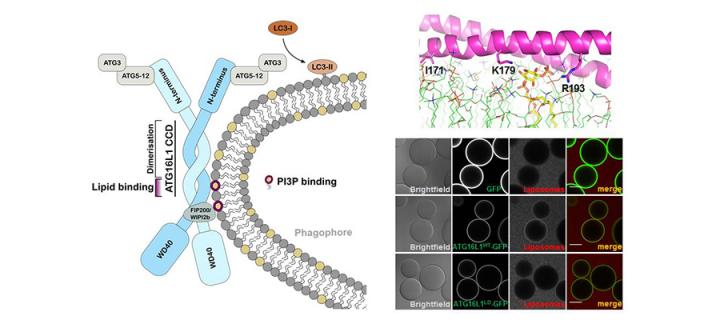
Cancer Research UK Edinburgh Centre scientists provide new insights into the regulation and function of ATG16L1 – a key autophagy protein: April 2019
Parliamentary Partnerships: From lab bench to back bench

Glasgow MP welcomed CGEM health data scientist to Parliament in Royal Society pairing scheme. April 2019
First research publication attributed to the XDF Programme
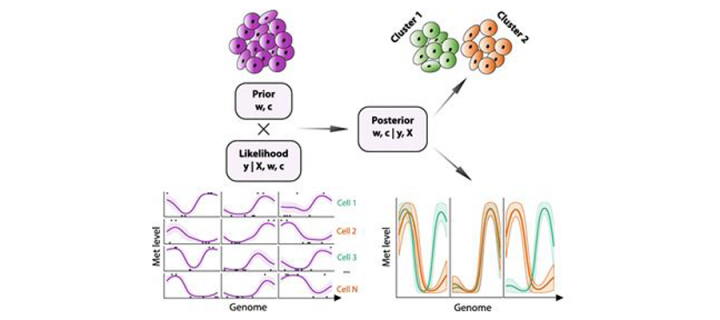
One of our XDF Programme Fellows, Dr Chantriolnt-Andreas Kapourani, authored an exciting research paper in “Genome Biology”: April 2019
Only a few days left to apply for cross-disciplinary fellowships

The application deadline for the MRC/UoE Cross-Disciplinary Post-Doctoral Fellowships (XDF) is 29 March 2019.
Science at the Interface to Industry

Workshop running from 24 - 26t April located at the Institute of Genetics and Molecular Medicine: March 2019
Edinburgh Cancer Discovery Unit featured in “SLAS Discovery”
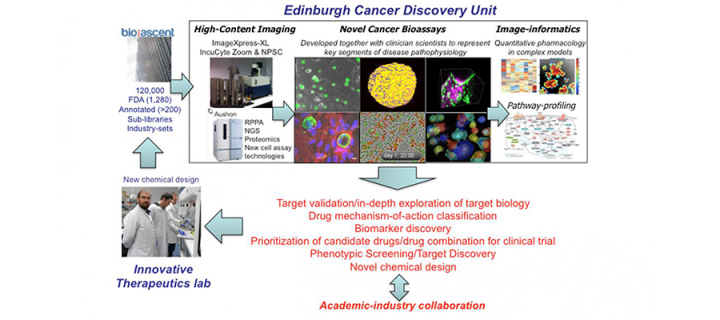
Editorial in a prominent drug discovery journal features CRUK Edinburgh Centre’s Edinburgh Cancer Discovery Unit and its phenotypic drug discovery activities: March 2019
Royal Society of Edinburgh Fellowship for David FitzPatrick

Fellowship recognises leadership in genetics of developmental disorders: March 2019
Shining a light on genetic disorders

The importance of the Patient Voice: February 2019
European Funding Success

Funding from the European Research Council will enable critical work on nanomedical devices designed to help treatment of cancer and chronic pain: February 2019
Study sheds light on damage linked to ageing

Some of the damaging cell effects linked to ageing could be prevented by manipulating tiny parts of cells, a study shows: February 2019
Finding the breaking point of cancer
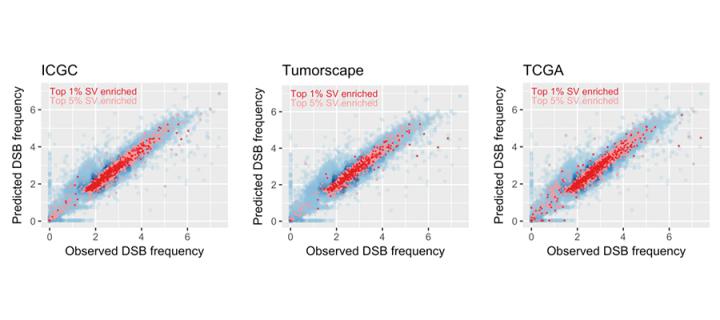
Cancer is a disease of the genome, and tumour sequencing projects have highlighted the enormous changes that the human genome undergoes during the evolution of a tumour: February 2019
HER2 drives an increased hypoxic response in breast cancer
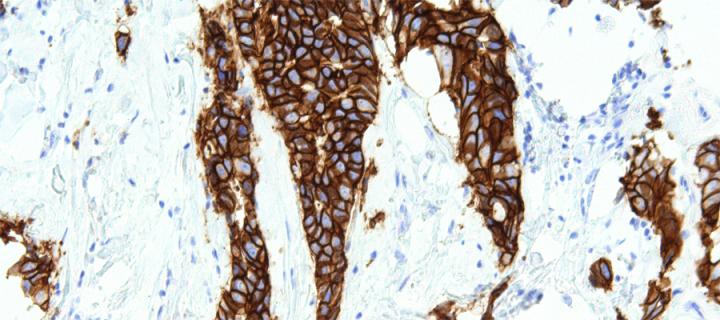
Cancer Research UK Edinburgh Centre scientists provide new insights into the role of HER2 in breast cancer cell signalling in hypoxia: February 2019
Developing and demonstrating public presentation skills

The IGMM Heat of the 3MT Competition and “Do Your Looks Betray You?”: February 2019
International student visit from the Zhejiang University-University of Edinburgh (ZJE) Institute, China Collaborations in Biomedical Sciences and Biomedical Informatics

On 24 January, the IGMM was delighted to welcome 14 undergraduate students from the Zhejiang University-University of Edinburgh (ZJE) Institute, China. They are in the third year of a four-year dual award programme in Integrative Biomedical Sciences: February 2019
Understanding the genetic basis of stress-sensitivity and depression.

In recent papers published in PLOS One and Translational Psychiatry, Dr Pippa Thomson describes work from her lab on the genetics of stress response and depressive symptoms. February 2019
Patients engage with research on Paget's Awareness Day

Professor Stuart Ralston’s Bone Group, in collaboration with the Paget’s Association, invited patients with Paget’s disease to attend a buffet lunch, followed with presentations on Friday 11: January 2019
Distinguishing acquired resistance from dormant tumours in neoadjuvant treatment of breast cancer

A study led by researchers from the Cancer Research UK Edinburgh Centre sheds new light on differences between dormant and treatment resistant tumours: January 2019
Women in Cell Biology Award for Pleasantine Mill

Pleasantine Mill honoured with the 2019 Women in Cell Biology Early Career Medal: December 2018
Reverse transcriptase inhibitors in Aicardi-Goutières syndrome
A study into the use of reverse transcriptase inhibitors for the treatment of Aicardi-Goutières syndrome. December 2018.

