Permeability imaging
Permeability imaging (dynamic contrast-enhanced MRI) involves the repeated acquisition of T1-weighted MR images following injection of a contrast agent such as gadolinium.
Overview
Purpose
Detail
Contact
- In the brain, the method can show quantitative information about the integrity of the blood-brain barrier (BBB).
- The BBB separates the parenchyma of the central nervous system from the blood. It regulates the passage of essential molecules while restricting the diffusion of potentially harmful substances.
- Disruption of the BBB can enable the extravasation of contrast agent, leading to an accumulation of contrast material in the extravascular extracellular space (EES). This results in increased longitudinal relaxation rate and, therefore, increased signal intensity in T1-weighted images.
- DCE-MRI exploits this T1 enhancement in order to detect and evaluate regions of BBB disruption. The repeated acquisition of T1-weighted images provides measurements of signal enhancement as a function of time, which differ between healthy and pathologic tissue (see Figure 1).
- The enhancement kinetics can be used to extract information regarding BBB integrity. Different approaches have been used to analyze DCE-MRI data, ranging from relatively simple visual assessment of enhancement curves to more complex fitting of pharmacokinetic models.
- Pharmacokinetic modelling can be used to relate the measured concentration-time curves to underlying physiological parameters relating to the contrast agent leakage. Pharmacokinetic modelling in DCE-MRI typically measures parameters relating to the permeability (permeability-surface area PS) and the volume of extravascular extracellular space (ve) and blood plasma space (vp), although with higher temporal resolution acquisitions it is also possible to gain information on tissue perfusion (blood flow Fp). An important physiological parameter that is widely reported in DCE-MRI studies is the volume transfer constant KTrans, which is the rate at which contrast agent is delivered to the EES per volume of tissue and contrast agent concentration.
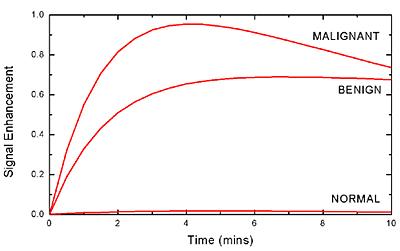
Figure 1. Typical signal enhancement "leakage profiles" characteristic of different tissue types in the brain, whereby malignant tumour tissue shows a high and fast enhancement followed by washout, benign tissue demonstrates a more progressive enhancement and normal tissue shows very little enhancement.
Research
Typical applications for DCE-MRI involve clinical disorders resulting in disruption of the blood-brain barrier. The most common imaging applications are in the study of focal lesions such as brain tumours (see Figure 2), multiple sclerosis plaques and acute ischaemic strokes. While these diseases show relatively large abnormalities in BBB functionality, there is also growing interest in the application of DCE-MRI to pathologies associated with more subtle and chronic BBB disruption, such as diabetes, cerebral small vessel disease, lacunar stroke (see Figure 3) and Alzheimer’s disease.
We are currently working with Anna Heye, a PhD student, whose project aims to improve the methodology of permeability imaging in small vessel disease and, thus, to move closer towards reliable quantification of subtle BBB abnormalities. Although there are many approaches for acquiring and analysing DCE-MRI data, these have mostly been developed for oncology applications, where vessel density and permeability are typically high. However, the optimal approach for detecting subtle BBB permeability is unclear. The small changes linked to subtle BBB breakdown are most likely influenced by secondary processes, such as random noise, scanner drift or partial volume effect. In order to enable reliable quantification of BBB integrity, it needs to be investigated whether the measured changes in the contrast-agent concentration curves are due to BBB disruption or other mechanisms. Furthermore, the project aims to find the most suitable approach for analysing DCE-MRI data related to subtle BBB disorders.
Recent and ongoing projects utilising this methodology include:
- Studies of permeability, perfusion and diffusion changes following dexamethasone treatment in high-grade intracranial tumours.
- Investigation into the contrast leakage related to small vessel disease and lacunar strokes
- Identification of leakage pattern distributions in a series of Alzheimer's disease patients compared to normal matched control subjects.
- Measurement of permeability, and diffusion changes following radiotherapy treatment in low grade intracranial tumours.
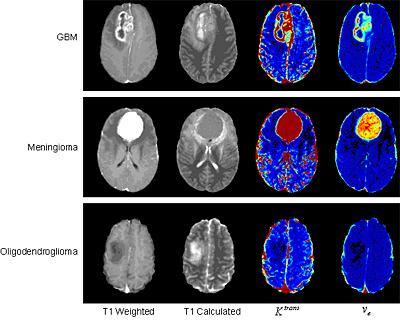
Figure 2. Example images obtained using DCE-MRI for three different types of intracranial tumour. Images shown are (from left to right) post-contrast T1-weighted, absolute T1, permeability-surface area product per unit volume of tissue (Ktrans) and extravascular extracellular space volume fraction (ve). The T1-weighted image is a standard clinical acquisition; the absolute T1 map gives a quantitative indication of tissue water content and is useful for assessing oedema; Ktrans indicates the endothelial permeability and ve shows the EES volume fraction, both of which provide an indication of blood-brain barrier integrity, which is clearly compromised more in the aggressive high grade tumours.
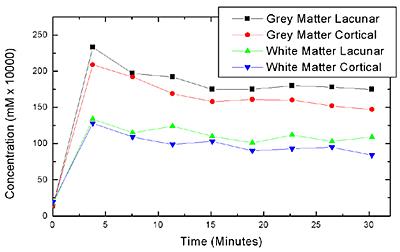
Figure 3. Preliminary data from 14 lacunar and 7 cortical stroke patients showing concentration-time curves obtained from grey and white matter regions of interest, which provide evidence that there may be a difference between the contrast agent accumulation properties in these different stroke sub-types.
Staff contact
If you wish further information on the above activities, please contact Dr Michael Thrippleton:

