Ian Kunkler: Clinical and Translational Studies of Breast Radiotherapy
Research Programme
Clinical /translational trials of adjuvant radiotherapy in early breast cancer
The BIG 2-04 MRC SUPREMO randomised trial with its administrative base at NHS Scotland (SCTRU) has recruited over 1600 patients with intermediate–risk breast cancer (1-3 positive axillary nodes/node negative with grade 3 histology/lymphovascular invasion) from UK, Europe (EORTC), Australia (TROG), Singapore and China.It closed to accrual in 2013 and is due to complete in 2023. It is supported by grants from the MRC, EORTC, Cancer Australia and the Trustees of the Hong Kong and Shanghai Bank. Its translational substudy (TRANS-SUPREMO (Prof John Bartlett,ECRC and Ontario for Cancer Research) is looking for molecular markers of breast cancer radiosensitivity and relapse. Thequality of life and health economic substudies are led by Professor Galina Velikova (Univ of Leeds) and Professor John Cairns (London School of Hygiene and Tropical Medicine, Univ of London).
The PRIME 2 trial, funded by the Chief Scientist’s Office for Scotland, randomised 1326 patients =/>65 years with hormone receptor positive, axillary node negative T1-2 (up to 3cm) after breast conserving surgery (1mm clear margins) receiving adjuvant hormonal therapy to whole breast radiotherapy (40-50Gy) or no further therapy. The 5 year local control rates were reported in 2015 (see publications).
The BIG 3.07 DCIS boost trial has randomised over 1600 patients with non low risk risk DCIS after breast conserving surgery and whole breast irradiation to a boost of irradiation to the site of excision or to no further therapy. A hypofractionated whole breast irradiation schedule (42.5 Gy in 16 fractions) is being compared to the standard dose fractionation regimes (50 Gy in 25 fractions). The trial is currently in follow up phase and will report in 2019. The trial is funded internationally by NHMRC in Australia (CI Assoc Prof Boon Chua) and in the UK by Breast Cancer Now and the Breast Cancer Institute, Edinburgh.
Translating the hypoxic microenvironment
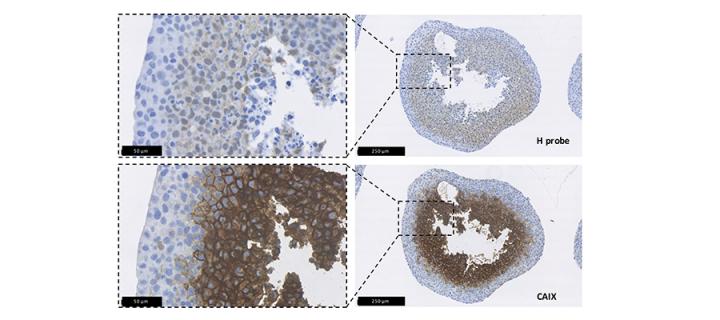
METOXIA is a 5 year, 22 partner EU FP 7 large scale integrated project completed in 2014. In collaboration with Professor David Harrison (St Andrew’s University), Dr Simon Langdon and Dr Carol Ward and Mr James Meehan (ECRC) and Prof Claudiu Supuran (Florence) a number of promising inhibitors of CA IX and NHEI have been evaluated in oxygenated and hypoxic conditions in preclinical models. Mr James Meehan is evaluating the role of the effects of targeting the pH regulators (CAIX, V-ATPase and NHEI) in preclinical models in normoxia and hypoxia. Additional support has been provided by the Breast Cancer Institute.
Sensing the tumour microenvironment
Hypoxia is an important contributory factor to resistance to radiotherapy and chemotherapy in breast cancer leading to relapse and death. At present we have no clinically applicable tools to capture the temporal and spatial changes in levels of oxygenation in solid tumours. If these changes could be measured, treatment by radiotherapy could be individualised, distributing higher doses using advanced radiotherapy delivery systems to hypoxic subregions to improve local control and survival.
In the current EPSRC funded (IMPACT project- Implantable Microsystems for personalised anti-cancer treatment), real time sensors (initially of oxygenation and pH) are being developed by Professor Alan Murray (CI) and Professor Anthony Walton (Electronic Engineering). Partners are Chemistry [Prof Mark Bradley, Prof Andy Mount) developing probes of cell death, Biology [Prof David Argyle, Roslin Institute, Dr Carol Ward, Dr Simon Langdon, Mr Mark Gray (ECRC)] testing the sensors, subject to necessary approvals, in murine and ovine models, Signal processing (Prof Steve McLaughlin, Heriot Watt University) and Value Systems (Prof Joyce Tait, Innogen Institute). I chair the clinical/translational committee to facilitate translation to ‘Ist in man’ studies of the sensors.

