Focal Adhesion Kinase activation on lipid membranes
Edinburgh researchers co-lead a study revealing how lipids activate the cancer protein Focal Adhesion Kinase via conformational changes: August 2020
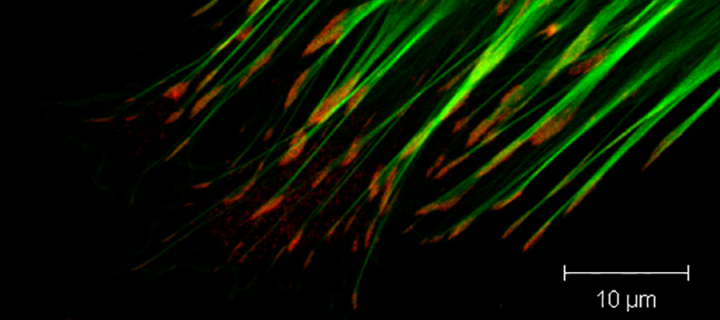
Focal adhesions are large protein complexes that assemble at the cytoplasmic side of the plasma membrane and connect the actin cytoskeleton inside the cell via integrin trans-membrane receptors to the extracellular matrix outside the cell. They play important role in transmitting mechanical force and regulatory signals between the extracellular matrix, controlling key cellular processes from cell survival and proliferation to cell motility and migration, and this signalling axis is hugely perturbed in cancer and other diseases.
Focal adhesions are highly dynamic and can contain over 100 different proteins, many of which have been recognised as potential oncogenes or tumour suppressors. Focal Adhesion Kinase (FAK) is one of the key components, or nodes, of focal adhesion complexes – building large signalling complexes in the vicinity of integrin-ECM attachment sites and at other cellular locales, like the nucleus. It both mediates cancer cell processes and buffers cancer cells from therapeutic insults; as such it is widely regarded as a good target for anticancer therapies and clinical trials with FAK inhibiotors are underway.
Despite the progress in elucidating FAK functions in the cells over many years, crucial aspects of its activation and protein complex-building at the membrane remain elusive. For example, in the cytosol, FAK adopts an autoinhibited state but is activated upon recruitment into focal adhesions at the membrane, yet how this occurs or what induces structural changes is poorly understood.
In a recently study, published in the EMBO Journal, entitled “Structural basis of Focal Adhesion Kinase activation on lipid membranes” researchers from the CRUK Edinburgh Centre, IGMM, worked in an international consortium with scientists from Spain, Switzerland, and Germany employing cryo-electron microscopy - an electron microscopy technique applied on samples cooled to very low temperatures and embedded in an environment of vitreous water - to reveal how FAK clusters into oligomers and associates with lipid membranes and how such membrane interactions unlock FAK autoinhibition to promote its activation. Linked to this, the Edinburgh group carried out mutational and biological analyses to validate the functional importance of the new model for FAK activation. These new insights will be vital in the design of novel agents targeting activation and specific functions of FAK. The study was published in the EMBO Journal and Professor Margaret Frame was a corresponding author.
Related Links:
Article in the EMBO Journal: https://www.embopress.org/doi/10.15252/embj.2020104743
Professor Margaret Frame group website: https://www.ed.ac.uk/cancer-centre/research/frame-group
What are focal adhesions?: https://www.mechanobio.info/what-is-mechanosignaling/what-is-the-extracellular-matrix-and-the-basal-lamina/what-are-focal-adhesions/
Interesting presentation about focal adhesions: https://www.youtube.com/watch?v=e9UXz8D-YWU
Related Stories:
Synergistic anticancer inhibitor combination discovered by a novel phenotypic screen:
Cancer immune evasion research on the cover of Science Signaling:
Edinburgh Cancer Discovery Unit featured in “SLAS Discovery”:
https://www.ed.ac.uk/cancer-centre/news-and-events/news-2019/ecdu-featured-in-slas-discovery
Development of new inhibitors of oncogenic receptor tyrosine kinases:

