A link between innate immune sensing and cellular senescence
Cancer Research UK Edinburgh Centre scientists discover a cell intrinsic tumour suppressor role of the innate immune sensor Toll-like receptor 2: June 2018
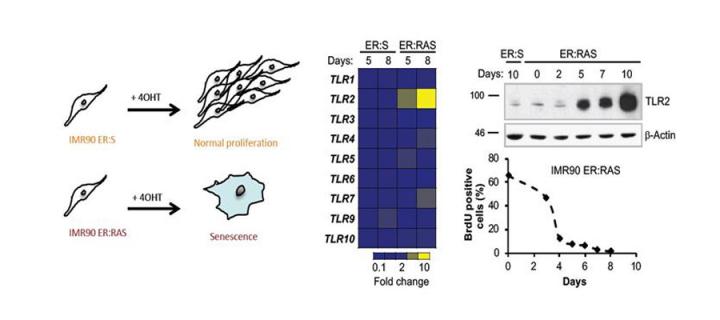
Cellular senescence is an anti-cancer mechanism characterised by a robust cell cycle arrest and the induction of a pro-inflammatory response. Cellular senescence limits the replicative capacity of cells, thus preventing the proliferation of cells that are at different stages of malignancy. A considerable body of evidence suggests that induction of senescence can be exploited as a basis for cancer therapy. On the other hand, accumulation of senescent cells in the body contributes to the acceleration of ageing and age-related diseases, and elimination of senescent cells or attenuation of senescence-associated pro-inflammatory response through senotherapies (therapeutic strategies specifically targeting cellular senescence) has been proven effective in combating age-related pathologies. Unfortunately, despite significant efforts, the molecular processes triggering and controlling cellular senescence remain elusive.
The immune system is a body defence mechanism that protects us against infectious organisms. To achieve this protection, cells of the immune system distinguish pathogens from the host by recognising foreign molecules through immune receptors and sensors. In a recent issue of “Science Advances” researchers from the Cancer Research UK Edinburgh Centre and their collaborators publish evidence that one of these receptors, Toll-like receptor 2 (TLR2), senses host molecules such as serum amyloids which are produced by cells undergoing oncogenic stresses, mediating a type of senescence that protects us against cancer. The study titled “The innate immune sensor Toll-like receptor 2 controls the senescence-associated secretory phenotype” was driven by Priya Hari, a PhD student, and led by Dr Juan Carlos Acosta from the “Cellular Senescence and Tumour Suppression” laboratory. Through defining a new senescence-associated pathway that regulates priming of inflammasome – a critical regulatory component of the innate immune response and the first line of defence against pathogens and damage – the authors provide exciting new links between innate immune signalling and senescence. They propose that cellular senescence shares mechanistic features with the activation of innate immune cells and could be considered a program of the innate immune response by which somatic cells switch their regular role to acquire an immune function under certain conditions of stress and danger, for instance upon oncogene activation. The results of the study not only extend our knowledge of molecular mechanisms controlling senescence but, in the future, may also lead to new cellular-senescence-based strategies for development of anticancer therapeutics or senotherapies based on innate-immune receptor manipulation.
Related links
Article in “Science Advances”: https://advances.sciencemag.org/content/5/6/eaaw0254
Juan-Carlos Acosta Group website: https://www.ed.ac.uk/cancer-centre/research/acosta-group
Learn more about cellular senescence in cancer: https://www.sciencedirect.com/science/article/pii/B9781455740666000159

