Synthesis of palladium activated analogue of anticancer drug irinotecan
Cancer Research UK Edinburgh Centre scientists synthesise bioorthogonally activated precursor of a potent topoisomerase I inhibitor: November 2018
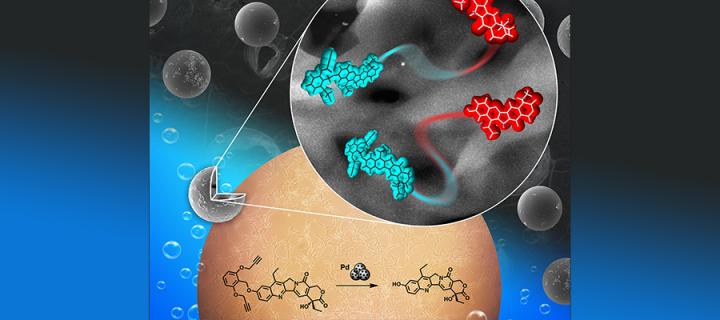
The Innovative Therapeutics Group led by Professor Asier Unciti-Broceta in the Cancer Research UK Edinburgh Centre are pioneers of applying gold and palladium nanoparticles for controlled release of anticancer drugs. Their strategy, known as bioorthogonal uncaging, enables transformation of harmless chemical compounds (prodrugs) into fully active anticancer therapeutics. It is designed to produce active therapeutic agents only in direct proximity of a tumour site, thus reducing harmful effects to other parts of the body. This is achieved with help of specially designed implants containing palladium or gold nanoparticles which control conversion of prodrugs into active drugs. Although these studies are still in preclinical phase they hold great promise for the future and the group has already engaged in collaborations with local clinicians including Dr Paul Brennan from the Centre for Clinical Brain Sciences.
In their recent study published in “Chemistry: A European Journal”, the Group described synthesis of the first inactive precursor of the potent topoisomerase I inhibitor SN38 that is specifically converted into active SN38 by palladium mediated chemical reaction. Cells need topoisomerase I enzyme to divide and grow, and SN38 is an active metabolite of the chemotherapy drug irinotecan used to treat many different types of cancer. Unfortunately irinotecan is converted into SN38 in the liver rather than at the tumour site and thus causes harm to many different tissues in the body before reaching the tumour site. The Group hopes that at some point in the future their new compound could pave the way to reducing adverse effects of irinotecan and similar therapeutics. Experiments performed using glioma and colorectal cancer cells in tissue culture confirmed effective conversion of their new compound into active SN38 in the presence of palladium nanoparticles and this caused death of many cancer cells. It was also possible to combine their new compound with Pro-5FU – a prodrug of another commonly used anticancer drug fluorouracil – to demonstrate for the first time the concomitant uncaging of two drugs used in clinical combinations by the same bioorthogonal method.
This work, featured as “Hot Paper” by the journal Editors, represents an important step in preclinical development of bioorthogonal organometallic approaches and may contribute to their transition into clinical trials.
Links
Article in “Chemistry: A European Journal”: https://onlinelibrary.wiley.com/doi/full/10.1002/chem.201803725
Innovative Therapeutics Group web page: https://www.ed.ac.uk/cancer-centre/research/unciti-broceta-group
Information about brain cancer: https://www.cancerresearchuk.org/about-cancer/brain-tumours
Information about colorectal cancer: https://www.cancerresearchuk.org/about-cancer/bowel-cancer
Information about nanoparticles: https://en.wikipedia.org/wiki/Nanoparticle
Information about irinotecan: https://www.cancerresearchuk.org/about-cancer/cancer-in-general/treatment/cancer-drugs/drugs/irinotecan

