Governance
Governance of ECMC's resources.
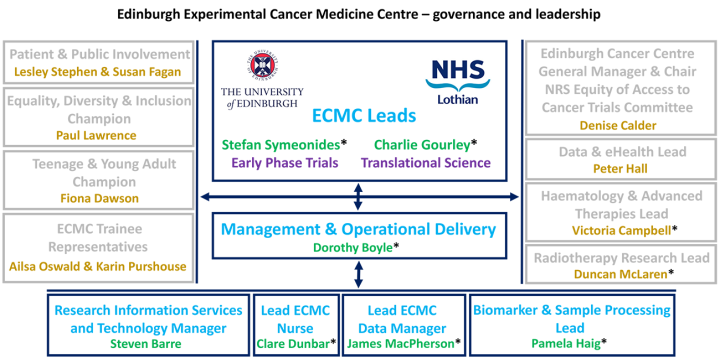
Edinburgh ECMC’s activities and resources are overviewed by the leadership team that includes:
• Co-lead (Early Phase Trials): Dr Stefan Symeonides
• Co-lead (Translational Science): Prof Charlie Gourley
• Haematology & Advanced Therapies: Dr Victoria Campbell
• Radiotherapy Research: Prof Duncan McLaren
• ECMC Management & Operational Delivery: Dorothy Boyle
• General Manager Edinburgh Cancer Centre & Chair National Research Scotland (NRS) Equity of Access to Cancer Trials Committee: Denise Calder
• Patient Public Involvement (PPI) Co-investigator: Lesley Stephen
• Patient Public Involvement (PPI) Coordinator: Susan Fagan
• Equality Diversity Inclusion (EDI) Champion: Paul Lawrence
• Teenage and Young Adult (TYA) Champion: Fiona Dawson
• Lead ECMC Nurse: Clare Dunbar
• Lead ECMC Data Manager: James MacPherson
• Biomarker & Sample Processing Lead: Pamela Haig
• Research Information Services & Technology Manager: Steven Barre
• Data and eHealth: Dr Peter Hall
• ECMC trainee representatives: Dr Ailsa Oswald and Dr Karin Purshouse

