Adele Marston
Orienting Chromosomes during Mitosis and Meiosis

Adele Marston is a Wellcome Investigator and Professor in Cell Biology at the University of Edinburgh. Her group aims to understand the origin of aneuploidy, particularly during meiosis, the cell division that generates eggs and sperm. The focus is to understand molecular mechanisms of chromosome segregation, primarily using yeast, together with a combination of genetics, biochemistry and microscopy.
Adele obtained her PhD from the University of Oxford, in the lab of Jeff Errington, and carried out postdoctoral work with John Chant at Harvard University and Angelika Amon at MIT. In 2005 she moved to the Wellcome Centre for Cell Biology in Edinburgh to establish her independent research group as a Wellcome Research Career Development Fellow. In 2010 she obtained a Wellcome Senior Fellowship, renewed in 2015. In 2021 she became a Wellcome Investigator and took over as Director of the Wellcome Centre for Cell Biology. She is an EMBO member (2019) and Fellow of the Royal Society of Edinburgh (2022).
Lab members
Eleanor Casey, Tiasha Ghosh, Marina Hamaia, Chuanli Huang, Dilara Kocakaplan, Lori Koch, Anett Ladanyi, Melanie Lim, Lucia Massari, Anuradha Mukherjee, Lucy Munro, Gerardus Pieper, Hollie Rowlands, Aparna Vinod, Lily Wang and Mansour Aboelenain
A simple explanation of research in the Marston lab - Research in a Nutshell Videos
Orienting Chromosomes during Mitosis and Meiosis
Specialization of chromosome segregation mechanisms in meiosis
Meiosis generates gametes with half the parental genome through two consecutive chromosome segregation events, meiosis I and meiosis II. Meiotic errors are prevalent in humans, accounting for frequent miscarriages, birth defects and infertility. Our vision is to elucidate the molecular basis of the adaptations that sort chromosomes into gametes during meiosis. We use budding and fission yeast as general discovery tools, and Xenopus and mouse oocytes to uncover meiotic mechanisms in vertebrates. Using patient-donated oocytes and ovarian tissue, we address the relevance of our findings for human fertility.
Structural and functional organisation of meiotic chromosomes
During meiosis, chromosomes undergo extensive remodelling for transmission into gametes. Chromosomes are broken and reciprocally exchanged in prophase, specifically cohered at centromeres during meiosis I and permanently separated at meiosis II. The cohesin complex is a major definer of chromosome structure, establishing intra and inter-sister chromatid linkages and providing the context for spatial control of homolog interactions. Cohesin defines a specialized chromosomal domain, called the pericentromere, surrounding each budding yeast centromere. We discovered that cohesin extrudes a chromatin loop on either side of the centromere until halted by convergent genes at pericentromere borders. Our recent work revealed that cohesin acetylation prevents extrusion through pericentromere borders and demonstrated that this boundary formation is critical for meiotic chromosome segregation. Therefore, we determined how chromosome loops are positioned to functionally structure the genome. A key ongoing focus is to understand how pericentromere structure influences its role as a signalling platform that safeguards chromosome segregation, both in the model yeast system and in human oocytes. In a new initiative, we are also using phospho-proteomics in synchronised yeast to uncover the cell cycle controls that allow two successive meiotic divisions.
Specialization of meiotic kinetochores
Kinetochores link centromeric nucleosomes to microtubules for chromosome segregation. Our goal is to understand how the kinetochore is adapted to perform its meiosis-specific functions in suppression of meiotic recombination, directing the co-segregation of sister chromatids during meiosis I, and maintaining linkages between sister chromatids until meiosis II. We defined the proteomic landscape of yeast kinetochores and centromeric chromatin during meiosis, revealing extensive remodelling during prophase and meiosis I. We are now addressing the mechanism of kinetochore remodelling, as well as its functional importance. In many organisms, sister kinetochores are fused in meiosis I, while a lack of fusion in human oocytes may account for susceptibility to segregation errors and fertility problems. Ongoing work in Xenopus, mouse and human oocytes aims to test this hypothesis.
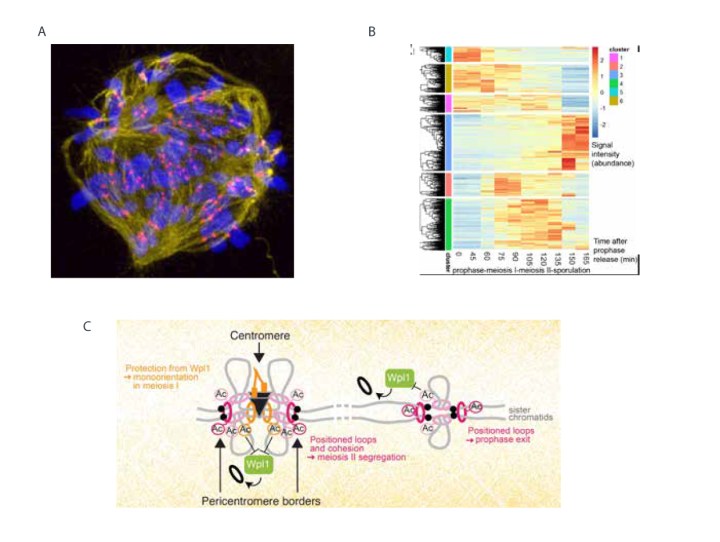
A. Mitosis 1 in the human embryo after inhibition of the CENP-E motor protein. The spindle is stained in yellow, centromeres in red and chromosomes in blue.
B. Phosphoproteome changes during budding yeast meiosis. Heatmap shows hierarchical clustering of changes in phospho-site intensity across a time course (meiotic prophase until sporulation).
C. Model showing how anchoring of chromatin loops and sister chromatid cohesion by Eco1-dependent acetylation of cohesin structures meiotic chromosomes for their segregation.
Selected publications
Barton R†, Massari LF†, Robertson D and Marston AL (2022). Eco1-dependent cohesin acetylation anchors chromatin loops and cohesion to define functional meiotic chromosome domains. eLife. 11, e74447. †Equal contribution
Su XB, Wang M, Schaffner C, Nerusheva OO, Clift D, Spanos C, Kelly DA, Tatham M, Wallek A, Wu Y, Rappsilber J, Jeyaprakash AA, Storchova Z, Hay RT and Marston AL. (2021) SUMOylation stabilizes sister kinetochore biorientation to allow timely anaphase. J Cell Biol. 220, e202005130.
Borek WE, Vincenten N, Duro E, Makrantoni V, Spanos C, Sarangapani KK, de Lima Alves F, Kelly DA, Asbury CL, Rappsilber J and Marston AL. (2021) The proteomic landscape of centromeric chromatin reveals an essential role for the Ctf19CCAN complex in meiotic kinetochore assembly. Current Biology 31, 283-296.

