Dr Jenna Cash
Jenna Cash's research focuses on understanding the events that determine whether a skin wound will heal acutely or develop into a chronic non-healing wound.
Dr Jenna Cash
Chancellor's Fellow and Sir Henry Dale Fellow

- Centre for Inflammation Research
Contact details
- Work: +44 (0)131 242 6655
- Email: jenna.cash@ed.ac.uk
- Web: Academic profile
- Web: Lab webpage
Group Members
-
Yonlada Nawilaijaroen - Research Technician
-
Valentina Moreno Mitchell
-
Yashna Rambeerich
-
Velizar Shinkov
-
Chistos Mavrommatis
- Dr Fraser Macrae (Sir Henry Wellcome Postdoctoral Fellow - joint with Professor R Ariens, Leeds University and Professor H Herwald, University of Lund)
-
Vignesh Jayaraman - PhD student (2nd supervisor with Dr Tovah Shaw)
-
Erqing Gao - PhD student (2nd supervisor with Prof Suhong Xu)
Background

The majority of skin wounds progress uneventfully through the sequential phases of healing, with the magnitude of the inflammatory response linked to the extent of the resultant scar. However, chronic wounds, including venous leg ulcers, diabetic foot ulcers and pressure ulcers, are thought to become ‘stuck’ in a chronically inflamed state, unable to heal effectively. Multiple immune and stromal cells, including macrophages, neutrophils, lymphocytes and keratinocytes play important roles in physiological and, most likely, pathophysiological healing. Macrophages are phenotypically plastic cells implicated both in supporting the ‘normal’ repair process, assisting angiogenesis, lymphangiogenesis and collagen deposition, and in playing detrimental roles in aberrant healing scenarios, with studies indicating that macrophages are dysfunctional, exhibiting unrestrained activation in the context of impaired healing in mouse and human. However, we lack a full appreciation of the dynamics of monocyte-macrophage recruitment and phenotype in acute skin wounds and macrophage biology has never been studied in a genetically tractable mammalian model of chronic wounds. An improved understanding of the acute repair program and how it becomes deranged to generate chronic wounds is urgently needed to inform new wound therapeutics. Exploiting resolution pathways may provide novel therapeutic strategies to stem the growing patient and healthcare burden that healing disorders inflict, perhaps by triggering resolution of chronic inflammation in non-healing wounds or reducing fibrosis.
Research Overview
We are interested in the events that influence whether a skin wound will heal acutely or develop into a chronic non-healing wound. We are focusing on the role of macrophages in determining wound outcome by addressing questions such as ‘how and when do macrophages become dysfunctional in aberrant healing scenarios’.
We use a variety of complementary approaches, including multiphoton live imaging of skin repair in mouse, flow cytometry and immunohistochemical techniques. We also take advantage of in vitro assays and human wound biopsies to help dissect mechanism and provide translational relevance.
Our second main research direction involves exploring whether pro-resolving pathways can be harnessed to drive healing, including rescue of chronic wounds, in order to develop novel therapies to improve healing and rescue chronic wounds.
The following PDF provides a brief visual summary of this group’s current research.
You can view a full catalogue of graphical research summaries for each group in the Centre for Inflammation Research by visiting our Research page.
Biographical Profile
Dr Jenna Cash graduated from the University of Edinburgh with a degree in Pharmacology (with Industrial Experience) in 2005. She was then awarded a British Heart Foundation DPhil studentship to identify novel endogenous anti-inflammatory pathways at the Sir William Dunn School of Pathology, Oxford University, with Professor David Greaves. Her DPhil research resulted in the discovery that a protein, chemerin, undergoes proteolytic processing by activated macrophages to generate peptides with dual anti-inflammatory and pro-resolving properties. Chemerin-derived peptides are under international patent and undergoing a clinical trial to treat skin inflammation. On completion of her DPhil, Dr Cash was awarded a Sir Henry Wellcome Postdoctoral Fellowship for 4 years, which she divided between Professor Mauro Perretti’s lab (William Harvey Research Institute, London), Professor Paul Kubes lab (Calgary University, Canada) and Professor Paul Martins lab (Bristol University). During this period she learnt specialised intravital microscopy techniques and developed a keen interest in wound healing, finding that chemerin peptide C15 can accelerate skin repair with reduced scarring and inhibit neutrophil integrin activation to reduce their recruitment to a site of inflammation. Dr Cash was subsequently awarded a 1 year Elizabeth Blackwell Early Career Research Fellowship by Bristol University/Wellcome ISSF, which provided her with the time and funds to pursue setting up her own lab. Dr Cash was able to establish a clinically relevant mouse model of human chronic wounds and multiphoton imaging of skin repair processes in the mouse. She ultimately secured a Chancellors Fellowship (University of Edinburgh) and a Sir Henry Dale Fellowship from the Wellcome Trust/Royal Society to establish her own lab within the MRC Centre for Inflammation Research at the University of Edinburgh in August 2016.
Honours and Awards
- Physiological Society Travel Award, 2015
- Emerging Investigator Prize, London Matrix Symposium, 2013
- Outstanding Young Investigator Award, William Harvey Research Institute Symposium, 2011
- Poster Prize at the World Congress on Inflammation, Tokyo, 2009
- BHF Centre of Research Excellence Travel Award, 2009
- Young Immunologist of the Year Award, British Society for Immunology Congress, Glasgow, 2008
Alumni
- Dr Holly Rocliffe
- Dr Antonella Pellicoro
- Swati Shaji
- Emma Guy
- Giulia Bartolomucci
- Dr Shani Austin-Williams
- Dr Georgios Krilis
- Ms Danielle Shields
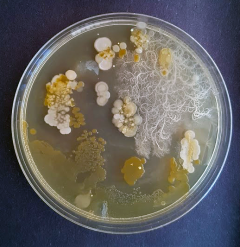
Public Engagement
As a Science, Technology, Engineering and Mathematics (STEM) Ambassador since 2010 I previously volunteered at public engagement events such as Centre of the Cell (London). On moving to the CIR, I have become involved in the Science Festivals subcommittee of our Public Engagement team as co-lead. I am particularly interested in discussing wound repair, including raising awareness of chronic wounds and visit primary schools in East Lothian to run ‘Meet the Microbes’ sessions, whereby the children grow their own microbial handprints on agar plates whilst learning of the importance of effective handwashing in relation to the role of microbes in disease.
Collaborators
Internal
- Dr Prakash Ramachandran (Centre for Inflammation Research, University of Edinburgh)
- Dr Andrea Caporali (Centre for Cardiovascular Research, University of Edinburgh)
- Professor Nick Casewell (Liverpool School of Tropical Medicine)
- Prof Julian Jones (Imperial University)
- Professor Zhihong Huang (Dundee University)
- Dr Michael Crichton (Heriott Watt University)
- Professor Jesmond Dalli (QMUL)
Sources of Funding
- Wellcome Trust and Royal Society
- Sir Henry Wellcome Postdoctoral Fellowship
- William Harvey Research Foundation
- Elizabeth Blackwell Early Career Research Fellowship
- Sir Henry Dale Fellowship
- Chancellors Fellowship
More information on funding at Jenna Cash's Research Explorer profile.

